
Introduction: Why Nutrition and Stomach Cancer Are Deeply Interconnected
Stomach cancer disrupts nutrition in uniquely devastating ways. Unlike other cancers, gastric cancer directly damages the organ responsible for digestion itself—blocking food passage, eliminating acid production, and destroying the stomach's elastic capacity. Malnutrition isn't merely a side effect; it drives poorer outcomes. Research shows that 75% of gastric cancer patients experience malnutrition at diagnosis, and this nutritional decline independently predicts reduced overall survival, increased surgical complications, and diminished tolerance to treatment.
The consequences cascade quickly. Stomach tumors cause mechanical obstruction, triggering early satiety, nausea, and vomiting long before treatment begins. Surgery worsens the crisis—severe malnutrition rates jump from 2.3% preoperatively to 26.3% post-gastrectomy, and patients may not regain baseline weight for an entire year.
This guide walks through each stage of nutritional intervention for gastric cancer:
- How the disease disrupts eating and nutrient absorption
- Dietary strategies during active treatment
- Managing post-surgery complications like dumping syndrome
- Supplementing critical nutrients after gastrectomy
- Using nutrition to actively support recovery, not just manage symptoms
TLDR
- Stomach cancer causes severe nutritional decline in up to 75% of patients; early intervention starting at diagnosis is essential to prevent complications and improve outcomes
- Small, frequent meals (5-6 daily) featuring soft, high-protein, calorie-dense foods help patients maintain weight despite reduced stomach capacity
- Avoid processed meats, salty/pickled foods, carbonated drinks, and high-fiber raw foods that increase digestive stress and worsen symptoms
- Post-gastrectomy patients require lifelong B12, iron, calcium, and folate supplementation due to impaired absorption
- Strategic nutritional therapy targeting cancer cell vulnerabilities, including methionine restriction combined with targeted nutraceuticals, may actively support treatment response
How Stomach Cancer Disrupts Nutrition — and Why Early Intervention Matters
Mechanical Obstruction and Symptom-Driven Malnutrition
Stomach tumors create physical barriers that block food from entering or leaving the stomach. This mechanical obstruction causes dysphagia (difficulty swallowing), early satiety (fullness after just a few bites), persistent nausea, and vomiting, drastically reducing caloric intake months before treatment starts. Patients struggle to eat enough simply because food cannot move through the digestive tract normally.
Cancer Cachexia: More Than Simple Starvation
Over 50% of gastric cancer patients develop cachexia, a metabolic syndrome characterized by involuntary weight loss, muscle wasting, and low albumin levels. Cachexia independently increases mortality risk by 40% (HR=1.397) in gastric cancer. Unlike starvation, cachexia accelerates energy expenditure even when intake drops: the body burns muscle and fat at abnormally high rates, driven by systemic inflammation and tumor-induced metabolic changes. Dietary intervention alone cannot reverse this without addressing those underlying mechanisms.

Preoperative Nutritional Status Determines Surgical Outcomes
Malnutrition before surgery worsens postoperative results in measurable ways. The Prognostic Nutritional Index (PNI), calculated as 10 × serum albumin (g/dL) + 0.005 × total lymphocyte count, identifies high-risk patients. Scores below 45 indicate severe impairment.
Low PNI predicts increased postoperative complications including infection, delayed wound healing, and hospitalization extending 6+ days beyond patients with adequate nutrition.
Preoperative enteral immunonutrition (enriched with arginine, omega-3 fatty acids, and RNA) for 7 days reduces infectious complications from 20% to 7.4% and anastomotic leak rates by half.
Post-Surgery Nutritional Deterioration
Malnutrition worsens sharply after gastrectomy. Body weight loss peaks at 6 months post-discharge, reaching approximately 8% below preoperative baseline. Many patients fail to regain their preoperative weight even 12 months after surgery, creating sustained nutritional deficits that impair healing and quality of life.
The Critical Role of Early Screening
ESPEN guidelines recommend nutritional assessment at diagnosis and repeated every 4-8 weeks. Validated tools identify at-risk patients before nutritional status becomes critical:
- NRS-2002 (Nutritional Risk Screening): Scores ≥3 predict higher postoperative morbidity (13.3% vs. 8.5%) and mortality (5.3% vs. 2.0%) in metastatic gastric cancer patients
- SNAQ (Short Nutritional Assessment Questionnaire): Scores ≥1 associate with 5-fold increased postoperative mortality risk
Early identification gives clinicians and patients a critical window to act: screening-guided intervention has been shown to reduce complication rates and shorten hospital stays in this population.
What to Eat: Dietary Principles for Stomach Cancer Patients
Soft, Easily Digestible Foods
When tumors partially obstruct stomach openings, nutrient-dense foods that pass easily become essential. Focus on:
- Soft proteins: Tofu, scrambled eggs, poached fish, plain yogurt, cottage cheese
- Healthy fats: Mashed avocado, olive oil drizzled on vegetables
- Calorie-dense condiments: Hummus, smooth nut butters, pureed soups
Avoid red meat and tough fibrous foods—these require extensive chewing and digestion that the compromised stomach cannot handle. Even soft, well-chosen foods need to be eaten in the right pattern to be tolerated.
Frequent Small Meals: 5-6 Times Daily
The stomach loses elastic capacity during cancer. Eating 5-6 small meals instead of 2-3 large ones is the primary strategy for meeting caloric and protein goals when portion size is limited.
Clinical guidance for post-gastrectomy patients recommends 6-8 small meals or snacks throughout the day to accommodate reduced capacity and prevent dumping syndrome.
Plant-Based and Whole-Food Choices
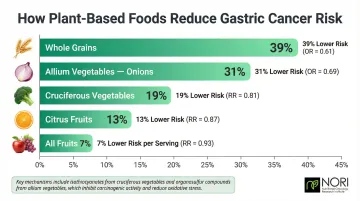
Fruits, vegetables, and whole grains each carry measurable protective effects against gastric cancer:
- Fruits: Higher intake reduces gastric cancer risk by 7% per serving (RR=0.93). Citrus fruits specifically lower risk 13% (RR=0.87)
- Cruciferous vegetables: High consumption decreases risk 19% (RR=0.81)—broccoli, cauliflower, Brussels sprouts contain isothiocyanates that activate detoxifying enzymes
- Allium vegetables: Onions reduce risk 31% (OR=0.69), garlic by 17%—sulfur compounds provide anti-inflammatory effects
- Whole grains: Consumption associates with 39% lower risk (OR=0.61)
Vitamin C scavenges reactive oxygen species and blocks carcinogenic nitrosamine formation; flavonoids add further antioxidant protection. When chewing is difficult, blend these foods into smoothies or soups to preserve the benefit.
Protein and Calorie Density
Patients who cannot maintain weight with food alone should use high-protein, high-calorie oral supplements between meals. Research shows oral nutritional supplements reduce body weight loss by 0.75 kg compared to dietary advice alone. Benefits last up to one year when intake exceeds 200 kcal/day.
Practical examples:
- Protein shakes made with nut butter, banana, Greek yogurt
- Commercial supplements (Ensure, Boost) between meals
- Smoothies with added protein powder and avocado
- Fortified soups with pureed beans
Separate Liquids from Solids
Drinking fluids during meals fills the reduced-capacity stomach too quickly, leaving no room for nutrient-dense foods. Drink high-calorie liquids 30-60 minutes before or after meals—not with them. Avoid carbonated drinks entirely to prevent gas buildup and bloating.
Foods and Habits to Avoid During Stomach Cancer Treatment
High-Sodium and Pickled/Smoked Foods
Salt intake significantly increases gastric cancer risk—high dietary salt raises risk 55% (OR=1.55), with a 12% increase per 5 g/day increment. Pickled foods increase risk by 32% (RR=1.32). These foods cause digestive discomfort and may promote disease progression.
Avoid: Soy sauce, canned soups, deli meats, pickled vegetables, smoked fish, salted crackers.
Processed and Red Meats
Western dietary patterns rich in processed meats increase gastric cancer risk 51% (OR=1.51). Processed meat consumption raises risk 24% (RR=1.24), with a 21% increase for every 50 g/day.
Mechanism: Nitrites in processed meats react with amines in the acidic stomach environment to form N-nitroso compounds, which cause DNA damage and cellular hyperproliferation. High nitrite intake increases risk 31% (RR=1.31).
Fried, Greasy, and Sugary Foods
Sugary foods trigger dumping syndrome: rapid gastric emptying that causes cramps, nausea, bloating, diarrhea, and dizziness. Fried and greasy foods create the opposite problem — they sit heavily in the reduced-capacity stomach, causing nausea and prolonged discomfort. Sugary beverages compound this by producing sharp blood sugar spikes followed by crashes.
Avoid: Fried foods, fast food, pastries, candy, sweetened beverages, desserts with added sugar.
Carbonated Drinks and Caffeine
Carbonation creates gas that bloats the reduced-capacity stomach. Caffeine irritates the stomach lining and accelerates gastric emptying, worsening dumping syndrome.
Avoid: Soda, sparkling water, energy drinks, coffee, strong tea.
High-Fiber Raw Foods
While fiber supports gut health, raw vegetables and whole grains take up stomach space and require extensive digestion. When stomach capacity is limited, prioritize calorie-dense foods over high-fiber raw options. Cook vegetables until soft to improve digestibility.
Avoid (or modify): Raw salads, cruciferous vegetables (eat cooked), whole grain bread, bran cereals, dried legumes.
Personalizing These Restrictions With a Dietitian
Some processed foods (like cereals that dissolve easily in milk) may be acceptable depending on individual tolerance and treatment type. Work with a dietitian to tailor these restrictions — what's off-limits for one patient may be fine for another.
Managing Common Eating Challenges
Dumping Syndrome
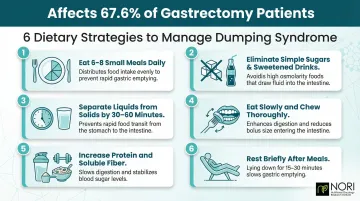
Dumping syndrome occurs when stomach contents move too quickly into the small intestine after gastrectomy, triggering cramps, nausea, bloating, diarrhea, and dizziness. Early dumping affects 67.6% of gastrectomy patients; late dumping affects 38.4%. Symptoms are most severe after total gastrectomy with Roux-en-Y reconstruction.
Dietary strategies to reduce dumping:
- Eat smaller, more frequent meals (6-8 times daily)
- Eliminate simple sugars and sweetened drinks—avoid fruit juice, candy, baked goods
- Separate liquids from solid meals by 30-60 minutes
- Eat slowly and chew thoroughly to slow gastric transit
- Increase protein and soluble fiber intake
- Lie down briefly after meals if advised
Early Satiety and Appetite Loss
Dumping syndrome is one of the most immediate post-surgical challenges — but early satiety often becomes the longer-term barrier to adequate nutrition.
Early satiety (feeling full after just a few bites) develops because tumors slow gastric emptying, and post-surgical changes to the stomach wall and vagus nerve compound this. Rapid nutrient delivery into the small intestine then triggers an exaggerated release of GLP-1 and GLP-2 hormones — both strong satiety signals — with hormonal spikes noticeably greater after total gastrectomy than distal gastrectomy.
Strategies to manage:
- Keep calorie-dense foods readily available—nut butters, cheese, crackers
- Eat by schedule rather than waiting for hunger
- Use appetite-stimulating foods (ginger, small amounts of citrus)
- Ask your physician about appetite-stimulant medications if persistent
- Seek mental health support—changed eating patterns affect emotional well-being and quality of life
For many patients, early satiety persists 6-12 months post-surgery. Building consistent meal timing habits early makes long-term adaptation significantly easier.
Essential Nutritional Supplements After Gastrectomy
Vitamin B12: Critical Deficiency Prevention
Incidence: Deficiency rates reach 100% after total gastrectomy within 4 years; median onset is 15 months post-surgery.
Why it occurs: Parietal cells that produce intrinsic factor are removed with the stomach. Intrinsic factor is essential for B12 absorption in the terminal ileum.
How to supplement: High-dose oral B12 (1,500 mcg/day) is as effective as intramuscular injections because approximately 1% of free B12 is absorbed via passive diffusion, bypassing the need for intrinsic factor. Alternatively, monthly B12 injections work well. ESPEN guidelines mandate annual B12 assessment via clinical symptoms and lab markers.
Iron: Preventing Anemia
Incidence: Anemia develops in approximately 50% of patients following total gastrectomy.
Why it occurs: Reduced gastric acid secretion impairs conversion of dietary nonheme iron (Fe3+) into absorbable ferrous form (Fe2+). Roux-en-Y reconstructions bypass the duodenum and proximal jejunum—the primary iron absorption sites.
How to supplement:
- Choose ferrous sulfate or ferrous gluconate over ferric forms for better absorption
- Take with vitamin C to enhance uptake
- Monitor hemoglobin and ferritin levels regularly
Calcium and Folate: Bone Health and Blood Formation
Duodenal bypass and altered gut anatomy reduce calcium and folate absorption after gastrectomy, raising the risk of osteoporosis and anemia. Both require active supplementation — diet alone is rarely sufficient.
Recommended approaches include:
- Calcium citrate: 1,200–1,500 mg daily in divided doses (better absorbed than carbonate without stomach acid)
- Folate: as directed by your provider based on lab values
- Comprehensive multivitamin: often prescribed when overall dietary absorption is unreliable
Important Caution
Supplements can interact with cancer treatments or cause unintended effects. Always consult your healthcare team before starting any supplement regimen. Select commercial nutrition supplements (protein shakes, meal replacements) with dietitian guidance to ensure they meet your specific needs.
Nutrition as Therapy: Moving Beyond Symptom Management
The Distinction: Support vs. Therapy
Nutritional support focuses on managing symptoms and preventing deficiencies during treatment—maintaining weight, replacing vitamins, easing digestive discomfort. Nutritional therapy takes a different approach: using dietary strategies to directly influence cancer cell behavior and the body's health-supporting response. Emerging research and integrative oncology programs recognize that what you eat can affect how cancer cells grow, divide, and respond to treatment.
Cancer Cells' Methionine Dependency
Cancer cells have markedly elevated methionine requirements compared to normal cells—a phenomenon called methionine auxotrophy or the Hoffman Effect. Methionine and cysteine are sulfur-containing amino acids essential for synthesizing glutathione, which cancer cells depend on to maintain elevated oxidative conditions, an area being actively investigated in nutritional oncology research. Restricting these amino acids produces a pro-oxidant state inside cancer cells — affecting cellular health pathways, as investigated in nutritional oncology research. Methionine modulation is being studied in nutritional oncology research for its role in cellular health and metabolic function in laboratory models.
Dietary methionine restriction — achievable through a predominantly plant-based diet that reduces animal protein intake — may selectively stress cancer cells while supporting normal cell function. Reducing animal protein in this way, without causing malnutrition, can support metabolic wellness, reduce IGF-1 levels, and is being explored alongside conventional care in ongoing research. Plant proteins (legumes, grains) contain far less methionine than animal proteins (meat, dairy, eggs). Adherence to a vegetarian diet associates with 45% reduced relative risk of developing stomach cancer (RR=0.55) compared to non-vegetarians in a 2026 pooled analysis of 1.8 million participants.
Clinical Evidence and Limitations
Moving from population data to clinical trials, the results are mixed. Early-phase trials show methionine-deficient parenteral nutrition synergizes with 5-fluorouracil chemotherapy to enhance tumor reduction in advanced gastric cancer patients. A Phase I trial combining a methionine-restricted diet with radiation, however, demonstrated poor feasibility—patients struggled to meet energy needs, resulting in unintended weight loss and leukopenia.
Important context: While epidemiological data strongly supports plant-based diets for preventing gastric cancer, no Phase III randomized controlled trial has proven that a low-methionine diet cures or directly treats established gastric cancer in humans. This remains an area of active investigation.
NORI Protocol: A Therapeutic Nutritional Framework
The Nutritional Oncology Research Institute (NORI) has developed a framework that applies methionine restriction therapeutically. Founded by Mark Simon, the NORI Protocol builds on over 20 years of focused research. It combines cycled methionine restriction—alternating periods of strict restriction with normal intake—with proprietary nutraceutical combinations targeting cancer cell vulnerabilities.
Where conventional nutritional support manages symptoms, the NORI protocol aims to actively support recovery and long-term management by:
- Restricting methionine and cysteine to no more than 10 mg/kg body weight daily through plant-based eating
- Using targeted pro-oxidant nutraceuticals (including sodium selenite, vitamin K3, genipin) that generate ROS or inhibit glutathione in cancer cells, affecting cellular health pathways in ways being investigated in nutritional oncology research
- Customizing protocols for individual cancer types and stages
- Continuously monitoring and adjusting the protocol as the patient's condition and response evolve
NORI's rationale for applying this framework broadly is that certain metabolic vulnerabilities—including methionine dependency and elevated oxidative stress thresholds—appear consistently across cancer types, not just gastric cancer. A free initial consultation is available for patients who want to explore whether this approach fits their situation (800-634-3804 or info@nutritionaloncology.net).
Frequently Asked Questions
What are the nutritional support options for stomach cancer patients?
Options range from oral dietary adjustments (small frequent meals, soft high-protein foods) to tube feeding when oral intake falls short, IV nutrition for those who can't tolerate either, and targeted vitamin and mineral supplementation after surgery. The right approach depends on cancer stage, treatment type, and surgical history.
What foods should stomach cancer patients avoid?
Avoid salty, pickled, and smoked foods; processed and red meats; sugary, fried, and carbonated foods and drinks; and high-fiber raw foods. These either increase digestive stress, worsen symptoms like dumping syndrome, or are associated with higher gastric cancer risk.
How do you manage dumping syndrome through diet?
Eat small, frequent meals (6-8 daily); avoid simple sugars and sweetened beverages; separate liquid and solid consumption at mealtimes by 30-60 minutes; and reduce portion size to prevent overfilling the post-surgical stomach.
What supplements are typically recommended after a gastrectomy?
Post-gastrectomy, impaired absorption makes B12, iron, calcium citrate, and folate essential — typically alongside a daily multivitamin. Your healthcare provider should guide specific choices based on lab results.
Can diet and nutrition affect stomach cancer outcomes?
Evidence links nutrition directly to outcomes: better pre-surgical nutritional status reduces complications and supports longer survival, while deficiency worsens prognosis. Diets rich in fruits, vegetables, and whole grains — and low in processed meats and sodium — are associated with the best results.
How does nutritional therapy differ from standard supportive care in stomach cancer?
Standard supportive nutrition focuses on maintaining weight and preventing deficiencies during treatment. Nutritional therapy, as practiced in integrative oncology, goes further by using targeted dietary interventions, such as methionine restriction and nutraceuticals, to directly influence cancer cell behavior and support recovery.


