
Introduction
Chronic diseases shape the global health landscape. As of 2023, 76.4% of US adults reported having at least one chronic condition, translating to 194 million individuals. Globally, the burden is even more severe: noncommunicable diseases killed over 43 million people in 2021, representing three-quarters of all non-pandemic-related deaths. Cardiovascular diseases account for over 19 million of these deaths annually, followed by cancers (10 million) and diabetes (1.6 million).
Diet drives this burden more than most realize. In 2017, dietary risks were responsible for 11 million deaths globally—22% of all adult deaths—making suboptimal nutrition one of the leading modifiable risk factors for chronic disease mortality. Diet, in other words, is not merely a lifestyle preference — it is a clinical tool capable of influencing disease progression, symptom burden, and long-term outcomes.
This article examines how nutrition affects chronic disease at a biological level, the evidence-based dietary strategies that show measurable clinical benefits, and why cancer patients stand to gain from targeted nutritional intervention.
For the Nutritional Oncology Research Institute (NORI) — whose founder Mark Simon has spent over 20 years researching nutritional therapies for cancer — food functions as medicine when applied with precision and scientific rigor.
TLDR
- 76% of US adults have at least one chronic disease
- 11 million global deaths annually are linked to dietary risk factors
- Evidence-based dietary patterns (Mediterranean, plant-based, low-glycemic) reduce risk for cardiovascular disease, diabetes, and cancer
- Cancer cells have unique metabolic vulnerabilities — including methionine dependence — that targeted nutrition can exploit
- Medical nutrition therapy delivers measurable clinical improvements across diabetes, CKD, and dyslipidemia
What Chronic Disease and Nutrition Actually Have in Common
The CDC defines chronic diseases as conditions lasting one year or more that require ongoing medical attention or limit daily activities. Cardiovascular disease, type 2 diabetes, metabolic syndrome, hypertension, chronic kidney disease, and cancer all qualify — and despite looking clinically distinct, they share a common upstream driver: poor dietary patterns.
Understanding how diet drives disease progression helps explain why nutritional intervention can influence so many conditions at once.
Biological Mechanisms Linking Diet to Disease
The connection between what we eat and chronic disease progression operates through several well-documented biological mechanisms:
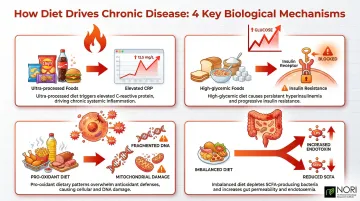
- Systemic inflammation — Higher ultra-processed food intake is associated with elevated hs-CRP levels, a key inflammatory biomarker. This chronic low-grade inflammation accelerates disease progression across cardiovascular disease, metabolic syndrome, and cancer.
- Dysregulated blood sugar — High-glycemic diets trigger insulin resistance. A dose-response meta-analysis linked high sugar-sweetened beverage consumption to a 0.21 increase in HOMA-IR among youth, setting the stage for type 2 diabetes.
- Oxidative stress — Pro-oxidant diets deplete antioxidant defenses and increase cellular damage markers that accelerate aging and tissue injury.
- Microbiome dysbiosis — Gut imbalance reduces short-chain fatty acid production, weakens gut barrier integrity, and increases endotoxins that fuel systemic inflammation. Research links dysbiosis to metabolic disorders, immune dysregulation, and cancer risk.
One chronic condition frequently feeds another. Obesity increases risk for type 2 diabetes, hypertension, and certain cancers simultaneously. This clustering effect is why diet functions as both a risk factor and a lever for change — modify one upstream driver, and multiple conditions can respond.
Diet-Related Conditions That Are Most Preventable
Certain chronic diseases respond most directly to dietary intervention:
- Obesity and metabolic syndrome — Caloric excess from ultra-processed foods drives weight gain and metabolic dysfunction; dietary modification remains the cornerstone of management.
- Type 2 diabetes — Healthful plant-based diets lowered type 2 diabetes risk by 30% (RR 0.70).
- Cardiovascular disease — The Mediterranean diet reduced major cardiovascular events by approximately 30% in the PREDIMED trial.
- Hypertension — The DASH diet reduced systolic blood pressure by 5.5 mm Hg more than control diets in foundational research.
- Certain cancers — Processed meat increases colorectal cancer risk by 18% per 50-gram daily portion, while plant-based diets reduce incidence.
The Global Burden of Disease Study estimated that high sodium intake, low whole grain consumption, and low fruit intake contributed to 255 million years of healthy life lost to disease globally. Most of that burden is preventable — which is exactly why dietary intervention deserves a central role in chronic disease management, not an afterthought one.
Evidence-Based Dietary Strategies for Chronic Disease Management
Not all dietary approaches are created equal. The strategies below are drawn from peer-reviewed clinical research — patterns with documented effects on inflammatory markers, blood sugar, cardiovascular risk, and cancer progression.
Anti-Inflammatory Eating
Reducing ultra-processed foods, red meat, trans fats, and refined sugars while increasing vegetables, fruits, legumes, and omega-3-rich foods lowers systemic inflammatory markers. Mediterranean dietary patterns reduce inflammatory markers including CRP and IL-6, which are implicated in cardiovascular disease and metabolic syndrome progression.
Plant-Based Dietary Patterns
Plant-based diets reduce incidence of hypertension, type 2 diabetes, and certain cancers through multiple mechanisms:
- High fiber content increases short-chain fatty acid production (particularly butyrate), improving gut barrier function and insulin sensitivity
- Phytonutrient content provides antioxidant and anti-inflammatory compounds
- Reduced saturated fat intake improves lipid profiles
- Lower IGF-1 signaling reduces cancer cell proliferation signals
Mediterranean and DASH Diets: Clinically Validated Patterns
These two dietary approaches have the strongest clinical evidence for chronic disease management:
Mediterranean Diet:
- Emphasizes olive oil, nuts, fish, vegetables, fruits, legumes, whole grains
- Reduced major cardiovascular events by 30% in the PREDIMED trial
- Supports cardiovascular and metabolic health through monounsaturated fats and polyphenols
DASH Diet (Dietary Approaches to Stop Hypertension):
- High in potassium, calcium, magnesium; low in sodium
- Reduced systolic blood pressure by 5.5 mm Hg and diastolic by 3.0 mm Hg more than control diets
- Specifically validated for hypertension management
Low-Glycemic Eating for Blood Sugar Management
Choosing foods with lower glycemic impact stabilizes insulin responses and supports diabetes management. Low-glycemic diets reduced HbA1c by a mean difference of -0.31% compared to higher GI control diets. This principle extends to cancer, where elevated blood sugar supports tumor cell metabolism through the Warburg effect (cancer cells' preference for glucose fermentation even in oxygen-rich environments).
What the Research Says About Dietary Patterns and Cancer Risk
That metabolic connection is just one of several pathways linking diet to cancer risk. The evidence across each is substantial:
- Plant diversity and fiber reduce carcinogenic compound exposure and lower colorectal cancer incidence through gut microbiome effects
- Processed meat is classified as a Group 1 carcinogen by the International Agency for Research on Cancer — each 50-gram daily portion increases colorectal cancer risk by 18%
- Excess body fat drives chronic inflammation and hormonal imbalances that raise risk for breast, colorectal, endometrial, and kidney cancers

Nutrition in Cancer Management: Beyond Prevention
Cancer is a chronic disease rooted in metabolic dysfunction, immune dysregulation, and cellular abnormalities. Nutritional intervention addresses several of these mechanisms directly — yet it remains one of the least integrated tools in oncology practice.
Exploiting Cancer Cell Metabolic Vulnerabilities
Cancer cells exhibit unique metabolic dependencies compared to healthy cells. One documented vulnerability is methionine dependence—many cancer cells cannot proliferate without external methionine supply, a phenomenon known as the "Hoffman effect." This dependency often arises from a defective methionine salvage pathway, frequently due to MTAP gene deletion.
Methionine Restriction Mechanisms:
Methionine restriction depletes S-adenosylmethionine (SAM), the body's primary methyl donor. This depletion:
- Inhibits histone and DNA methylation in cancer cells
- Disrupts redox homeostasis
- Has been investigated for its effects on cellular stress pathways, including cell cycle regulation in tumor cells
Preclinical models demonstrate that methionine restriction is being researched for its role in metabolic health and cellular function across multiple cancers. A landmark 2019 human feasibility study published in Nature showed that healthy participants on a low-methionine diet achieved a 60% reduction in serum methionine levels with good tolerability.
Normal cells adapt to methionine restriction through metabolic flexibility, while cancer cells—lacking this adaptability—face selective stress.
The Anti-Cancer Role of Plant-Derived Compounds
A predominantly plant-based, whole-food diet creates a biochemical environment less hospitable to cancer growth through:
- Polyphenols and flavonoids that modulate cell signaling pathways
- Sulfur-containing phytonutrients (from cruciferous vegetables) that activate detoxification enzymes
- Fiber that promotes beneficial gut microbiota and reduces systemic inflammation
Nutrition During Cancer Treatment
Beyond targeting cancer cells directly, nutrition during treatment:
- Supports immune function
- Reduces treatment side effects
- Preserves lean body mass
- Maintains quality of life
Conventional treatment protocols rarely integrate nutrition as a core component — a gap that organizations focused on nutritional oncology have worked to fill.
NORI's Approach to Nutritional Oncology
The Nutritional Oncology Research Institute (NORI) has researched nutritional interventions for cancer since 2004. Their home-based program centers on cycled methionine restriction combined with targeted nutraceutical support, designed to complement a patient's existing treatment plan — with no clinic visits required.
One documented case illustrates the approach: a Stage IV papillary thyroid cancer patient achieved a thyroglobulin reduction from 6.0 to 0.25 within two months using methionine restriction combined with sodium selenite and vitamin E delta tocotrienol. NORI has observed similar responses in breast and prostate cancer cases.
NORI formulates and manufactures all nutraceuticals at its own facility, maintaining direct control over quality standards and research-driven formulation decisions.

The Role of Nutraceuticals and Targeted Supplementation
Nutraceuticals are concentrated bioactive compounds derived from food or plant sources that, when used strategically, support immune function, reduce inflammation, and interact with disease pathways. They differ from general vitamins (which address deficiencies) and pharmaceuticals (which are synthetic drugs).
Research-Supported Nutraceuticals
| Nutraceutical | Target Condition | Clinical Evidence | Mechanism |
|---|---|---|---|
| Omega-3 Fatty Acids (EPA) | Cardiovascular Disease | Icosapent ethyl reduced ischemic events by 25% (HR 0.75) | Modulates inflammatory cascades and endothelial function |
| Berberine | Metabolic Syndrome/Type 2 Diabetes | Reduces triglycerides by 23.70 mg/dl, LDL by 9.63 mg/dl, HbA1c by 0.45% | Improves glucose and lipid metabolism |
| Sulforaphane | Cancer Biology | Has been investigated for its effects on cellular stress pathways in preclinical models; early trials support safety | Activates Nrf2-ARE antioxidant pathways; inhibits HDACs |
| Curcumin | Cancer Biology | Downregulates PI3K/Akt signaling in tumor cells | Modulates NF-κB pathways, enhances immune targeting |
Applying this research to cancer-specific protocols requires more than selecting well-studied compounds — it demands formulations calibrated to how cancer cells behave metabolically. NORI develops and manufactures its own nutraceutical products in-house, pairing them with the methionine restriction dietary protocol to exploit cancer cells' dependence on the amino acid methionine.
Their formulations include compounds selected for their role in oxidative stress pathways and cellular health being investigated in nutritional oncology research. These pro-oxidant agents are being studied for their interaction with oxidative stress conditions associated with cancer cells, with ongoing research into their selective effects. Cancer cells are known to have elevated ROS levels; these agents are being investigated for how they may interact with these cellular conditions. Natural pro-oxidant agents used include high-dose Vitamin C, piperlongumine, menadione, and certain carotenoids, which are also being explored alongside conventional care approaches in ongoing nutritional oncology research:
- Sodium selenite — being studied for its role in cellular health pathways in nutritional oncology research
- Vitamin K3 — a pro-oxidant agent in cancer cells that generates oxidative stress and depletes glutathione
- Genipin (gardenia fruit extract) — disrupts mitochondrial function in tumor cells
- Vitamin E delta tocotrienol — inhibits tumor cell proliferation and angiogenesis
Each compound is chosen based on peer-reviewed evidence for its mechanism of action against specific cancer cell vulnerabilities, particularly those exposed by methionine restriction.
Medical Nutrition Therapy: How Structured Nutritional Care Works
Medical nutrition therapy (MNT) is a personalized, evidence-based clinical process administered by qualified nutrition professionals, typically Registered Dietitian Nutritionists (RDNs). MNT involves:
- Formal nutrition assessment
- Individualized intervention plan based on medical history, comorbidities, medications, and cultural preferences
- Ongoing monitoring and adjustment
Conditions Where MNT Shows Strong Clinical Evidence
- Diabetes: MNT provided by RDNs produces HbA1c decreases up to 2.0% in type 2 diabetes within 3-6 months — one of the strongest dietary intervention outcomes on record.
- Chronic Kidney Disease: Sodium-specific MNT reduced systolic blood pressure by 6.7 mm Hg and lowered urinary sodium excretion in non-dialyzed CKD patients.
- Dyslipidemia: Multiple MNT sessions with a dietitian improved total cholesterol, LDL-C, and triglycerides compared to usual care, with measurable healthcare cost savings.
- Cancer-Related Malnutrition: Systematic reviews indicate MNT improves quality of life, supports weight preservation, and reduces unplanned hospital admissions in oncology patients.

Generic Advice vs. Structured MNT
The difference comes down to individualization. Generic dietary advice applies broad population-level guidelines — structured MNT accounts for:
- A patient's complete medical profile and active medications
- Nutrient-drug interactions that can affect treatment
- Cultural food preferences that directly impact adherence
- Readiness to change, which shapes how interventions are sequenced
This tailored approach consistently produces better clinical outcomes than one-size-fits-all recommendations.
Building Nutritional Habits That Last
Changing long-standing food habits is hard — especially for cancer patients managing symptoms, treatment side effects, and the emotional weight of a serious diagnosis. When expectations are too ambitious from the start, adherence breaks down before results have a chance to appear. Small, structured changes tend to outlast dramatic overhauls.
Practical Strategies for Long-Term Adherence
Setting goals that stick:
- Focus on incremental changes rather than complete dietary overhauls
- Set specific, measurable targets (e.g., "add two servings of vegetables daily" rather than "eat healthier")
- Acknowledge small wins — they sustain motivation through the harder stretches
Tracking progress:
- Monitor concrete indicators like weight, blood sugar, or energy levels
- Dietary self-monitoring correlates with greater weight loss, though paper logging loses traction over time
- Digital tools reduce the logging burden while preserving accountability
Building a support network:
- Establish accountability partnerships with family, friends, or health coaches
- Connect with communities that share similar health goals
- Motivational interviewing improves diet quality and supports weight loss, with the strongest outcomes in programs lasting at least 6 months
Adherence in Cancer Patients
For cancer patients specifically, adherence closely tracks with how manageable the protocol feels day-to-day. NORI's program is built around home-based participation, so patients avoid clinic visits while receiving unlimited support and ongoing guidance. The organization offers free weekly online Zoom workshops where patients can hear testimonials, ask questions, and find community support—reinforcing adherence through peer connection.
Frequently Asked Questions
What is medical nutrition therapy for chronic disease?
Medical nutrition therapy is a clinical, personalized nutrition plan developed by a registered dietitian to help prevent, manage, or treat specific chronic conditions through evidence-based dietary changes and ongoing monitoring.
Can nutrition alone manage a chronic disease like cancer?
While nutrition is a powerful tool, it is most effective as part of an integrative approach. For cancer patients, nutritional intervention can meaningfully support outcomes but typically works best alongside other elements of a comprehensive care plan.
What is methionine restriction and why does it matter for cancer patients?
Methionine is an amino acid abundant in animal proteins that cancer cells depend on more heavily than healthy cells. Restricting it through a primarily plant-based diet may help create conditions less favorable to cancer cell survival while normal cells adapt.
What dietary pattern is best for reducing chronic disease risk?
No single "best" diet exists for everyone, but anti-inflammatory, plant-rich dietary patterns—such as the Mediterranean diet or whole-food plant-based eating—consistently show the strongest evidence across multiple chronic disease categories.
How does diet affect inflammation and chronic disease progression?
Certain foods, particularly ultra-processed items, refined sugars, and trans fats, promote systemic inflammation: a core mechanism in conditions from cardiovascular disease to cancer. Whole-food, plant-based diets reduce inflammatory markers and slow disease progression.
Are nutraceuticals safe to use alongside conventional chronic disease treatments?
Many nutraceuticals are well-tolerated and research-supported, but patients should work with a knowledgeable practitioner to ensure there are no interactions with medications or treatments. Quality and sourcing matter significantly.
What we eat shapes disease trajectories in measurable, meaningful ways — from cardiovascular risk to cancer progression. For patients looking to augment their treatment plans, adopting evidence-based dietary patterns and working with qualified nutrition professionals offers a well-supported path forward. NORI's work shows how targeted nutritional intervention, applied with scientific rigor, can complement conventional care and support recovery across even advanced cancers.


