
Introduction
Between 20% and 85% of cancer patients use dietary supplements after diagnosis, yet most do so without professional guidance — shopping from health store shelves or online vendors, relying on marketing claims rather than biological mechanisms.
This pattern of unguided supplementation carries real risk. The evidence landscape for nutraceuticals in oncology is far more conditional than supplement labels suggest: some compounds show genuine health-supporting activity at specific doses, while others may inadvertently support tumor growth when used incorrectly.
This article provides a clear, evidence-based look at what nutraceuticals actually are, how they work biologically, what clinical research shows (and doesn't show), and how to use them responsibly as part of a cancer care strategy. The stakes are real: 2024 biphasic dosing findings and documented drug interaction risks both show that good intentions without proper guidance can produce harmful outcomes.
TL;DR
- Nutraceuticals are bioactive compounds from food sources that influence cancer-related biological pathways such as oxidative stress, chronic inflammation, and immune evasion
- Several well-studied nutraceuticals—curcumin, vitamin D, omega-3s, and sulforaphane—show documented health-supporting effects and can reduce chemotherapy side effects at the right doses
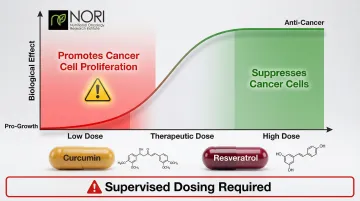
- Dosing precision is critical: sublethal doses of some nutraceuticals (curcumin, resveratrol) can promote cancer cell growth rather than suppress it
- Clinical evidence is strongest for vitamin D (improved pathologic complete response in breast cancer), probiotics (reduced chemoradiotherapy diarrhea), and omega-3s (reduced treatment toxicity)
- Work with a knowledgeable practitioner before starting any regimen — nutraceuticals can alter drug metabolism and interfere with active treatment
What Are Nutraceuticals, and How Do They Work Against Cancer?
Defining the Category
Nutraceuticals are bioactive compounds derived from food sources that provide health benefits beyond basic nutrition. This category includes vitamins (D, C, E), minerals (selenium), polyphenols (curcumin, resveratrol, quercetin), herbal extracts (green tea EGCG), probiotics (Lactobacillus species), and fatty acids (omega-3s). They differ from functional foods (whole foods with health benefits) and pharmaceuticals (synthetic drugs requiring FDA approval).
In the U.S., nutraceuticals are regulated as dietary supplements under the Dietary Supplement Health and Education Act (DSHEA) of 1994. Unlike pharmaceuticals, manufacturers are not required to demonstrate safety or efficacy before selling — a regulatory gap that creates real quality and safety concerns for patients navigating this space.
The Biological Problem
Cancer's uncontrolled growth is driven by several interconnected mechanisms at once:
- Chronic oxidative stress damages DNA and fuels cancer initiation
- Systemic inflammation creates a tumor-supportive environment
- Epigenetic dysregulation silences tumor suppressor genes
- Immune evasion allows cancer cells to escape detection
- Aberrant signaling pathways drive proliferation and metastasis
Unlike most chemotherapy drugs that target a single pathway, nutraceuticals can modulate multiple mechanisms simultaneously. That broad reach matters — cancer rarely fails for just one reason.
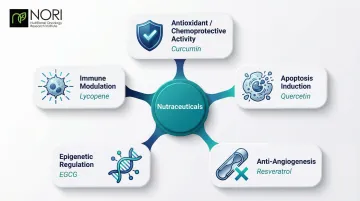
Key Anticancer Mechanisms
Antioxidant and Chemoprotective Activity: Oxidative stress—caused by reactive oxygen species (ROS)—damages cellular DNA and accelerates cancer progression. Antioxidant nutraceuticals like curcumin activate the Nrf2 pathway, which turns on genes for phase II detoxification enzymes. Quercetin and lycopene scavenge ROS, protecting healthy cells from chemotherapy's cytotoxic collateral damage. Cancer cells, already operating under elevated oxidative stress, are being investigated for how they may interact with these cellular conditions—an area of active nutritional oncology research.
Direct Cellular Health Mechanisms: Several nutraceuticals have been investigated for their effects on cellular stress pathways through specific mechanisms:
- Curcumin has been studied for its effects on cellular stress pathways and regulation of phase-II antioxidant enzymes
- Quercetin inhibits the PI3K/Akt pathway, being researched for its role in metabolic health and cellular function in breast, prostate, pancreatic, colon, and brain cancers
- Sulforaphane suppresses epithelial-to-mesenchymal transition (EMT) by downregulating ZEB1 and Snail, being studied for its role in cellular health
These compounds also block angiogenesis (new blood vessel formation that feeds tumors) by inhibiting VEGF expression, and suppress proliferation through NF-κB and MAPK pathway interference.
Epigenetic and Immunomodulatory Mechanisms: Some nutraceuticals work at the gene-expression level. Genistein (from soy) and EGCG (from green tea) act as DNA methyltransferase (DNMT) inhibitors — removing methyl groups from silenced tumor suppressor genes and switching them back on. Resveratrol enhances CD8+ T cell and natural killer (NK) cell cytotoxicity while reducing regulatory T cells that dampen immune responses. Curcumin modulates pro-inflammatory cytokines (IL-6, TNF-α) directly within the tumor microenvironment.
Taken together, these mechanisms explain why researchers are investigating nutraceuticals not just as adjuncts to conventional treatment, but as foundational tools in long-term cancer management.
The Most Studied Cancer-Fighting Nutraceuticals
Curcumin (Turmeric)
Curcumin is the most clinically studied nutraceutical in oncology, tested in over 100 clinical trials. It targets multiple pathways simultaneously—anti-inflammatory through NF-κB inhibition, pro-apoptotic through p53 activation, and anti-angiogenic through VEGF suppression.
The Bioavailability Problem: Phase I trials show curcumin is safe up to 8,000 mg/day, but standard oral doses yield only nanomolar plasma concentrations—far below the 1-100 µM needed for health-supporting activity in cell studies. In one trial, no curcumin was detected in serum at doses up to 8,000 mg; only at 10,000-12,000 mg were low levels measurable.
Solutions: Combining curcumin with piperine (black pepper extract) increases bioavailability by 2000% in humans. BCM-95 formulations (containing essential oils/lipids) provide 6.93-fold higher bioavailability than standard curcumin, while nanoparticle formulations like Theracurmin achieve 27-fold higher area-under-curve concentrations.
Resveratrol (Grapes, Berries)
Resveratrol inhibits PI3K/AKT, STAT-3, and NF-κB pathways, induces G1-phase cell cycle arrest, and enhances NK cell activity.
Biphasic Warning: A 2024 Journal of Clinical Oncology abstract found that at sublethal doses—corresponding to typical supplement consumption levels—both curcumin and resveratrol actually promoted cancer cell proliferation in vitro. This biphasic dose-response pattern means neither compound should be self-administered without clinical oversight to confirm therapeutic dosing ranges.
Vitamin D
Vitamin D carries some of the strongest RCT evidence of any nutraceutical in oncology.
RCT Evidence: A 2025 prospective RCT found that weekly 50,000 IU oral vitamin D3 supplementation during neoadjuvant chemotherapy significantly improved pathologic complete response rates in breast cancer patients, particularly those with hormone receptor-negative tumors (OR: 2.33, 95% CI 1.20-4.53). Vitamin D is also linked to improved recurrence-free survival in adenocarcinoma patients with low baseline levels—making baseline testing a practical first step for any cancer patient.
Sulforaphane and EGCG
Sulforaphane (from broccoli, cruciferous vegetables) inhibits histone deacetylase (HDAC) activity to restore pro-apoptotic gene expression, suppresses EMT to limit metastasis, and enhances phase II detoxification enzymes (enzymes that neutralize carcinogens). Preclinical models show synergistic potential with cisplatin.
The Culinary Detail: Cooking destroys the myrosinase enzyme required to convert glucoraphanin into bioactive sulforaphane. However, adding powdered brown mustard (an exogenous myrosinase source) to cooked broccoli increases sulforaphane bioavailability by over four times.
EGCG (green tea catechin) inhibits proteasome activity and modulates epigenetic machinery as a DNMT inhibitor. Combined with sulforaphane, EGCG enhances cisplatin-induced apoptosis in both cisplatin-sensitive and cisplatin-resistant ovarian cancer cells.
Omega-3 Fatty Acids and Probiotics
Of all nutraceuticals studied, omega-3 fatty acids and probiotics have the most consistent RCT evidence for reducing treatment side effects—not just theoretical health-supporting mechanisms, but measurable symptom relief during active therapy.
Omega-3s: A 2023 umbrella review of meta-analyses found omega-3 supplementation significantly reduced tumor necrosis factor alpha (TNF-α) (SMD: −0.34) and interleukin-6 (IL-6) (SMD: −0.30) in gastrointestinal cancer patients. Systematic reviews show reduced treatment toxicity when omega-3s are incorporated into chemotherapy regimens.
Cooking matters: Frying destroys EPA and DHA through oxidation at high temperatures and exchange between food and cooking oils. Baking or light steaming preserves omega-3 content better.
Probiotics: A 2025 meta-analysis of 18 RCTs found probiotic supplementation significantly reduced chemoradiotherapy-induced diarrhea in colorectal cancer patients (RR=0.51; 95% CI: 0.40-0.64). For patients undergoing colorectal chemoradiotherapy, this translates to a roughly 50% reduction in diarrhea risk—one of the most debilitating and treatment-limiting side effects in this population.
What Clinical Research Actually Shows
The Translation Gap
Preclinical data (cell and animal studies) for nutraceuticals is extensive, but clinical translation has been inconsistent. Studies suffer from variable designs, small sample sizes, short follow-up periods, and diverse patient populations.
Curcumin illustrates this gap clearly. Safe up to 8g/day in Phase I trials and showing reduced NF-κB and COX-2 in several studies, yet a Phase II RCT combining curcumin and anthocyanins in adenomatous polyp patients showed no significant change in inflammatory biomarkers.
The Biphasic Dosing Warning
A 2024 Journal of Clinical Oncology abstract tested 6 widely marketed nutraceuticals (curcumin, resveratrol, rosemary extract, rosmarinic acid, hederagenin, kava extract). While high doses suppressed cancer cells, sublethal low doses—which correspond to typical supplement consumption levels—actually promoted cancer cell proliferation in vitro.
Clinical trials with these compounds showed short follow-up and small sample sizes, with most showing negative or minimal health-supporting effects. Critically, no trials were designed to detect pro-growth effects. This finding demonstrates why "more is not always better" and unsupervised supplementation carries genuine risk.
Where Evidence is Strongest
Some nutraceuticals have accumulated enough clinical evidence to inform real treatment decisions:
- Vitamin D — associated with improved pathologic complete response and survival in breast cancer when supplemented during neoadjuvant therapy
- Probiotics — RCT data supports reduced chemoradiotherapy side effects, particularly diarrhea in colorectal cancer patients
- Omega-3 fatty acids — meta-analytic evidence for reduced treatment toxicity and improved inflammatory markers
- Ginger — meta-analysis-supported efficacy for chemotherapy-induced nausea
These four represent the clearest starting points for patients and clinicians weighing integrative support options.
The Nutrigenomics Frontier
Individual genetic variations mean the same nutraceutical can produce very different outcomes in different patients. MTHFR gene polymorphisms affect folate metabolism and may predict response to immune checkpoint blockades. Vitamin D receptor (VDR) gene SNPs influence vitamin D efficacy in prostate and colon cancer patients.
This field — nutrigenomics — is reshaping how researchers think about personalized nutritional protocols in oncology. That said, MTHFR testing, though widely used, may not provide reliable disease risk estimates in populations with significant genetic admixture, limiting its current clinical utility.
Nutraceuticals as Adjuncts to Conventional Cancer Treatment
Synergy Potential
Nutraceuticals are being explored alongside conventional care in ongoing research by targeting complementary molecular pathways and affecting how cancer cells respond to treatment. Curcumin is being explored alongside conventional care in ongoing research with 5-fluorouracil, doxorubicin, and cisplatin partly by inhibiting pro-survival signaling and downregulating multidrug resistance proteins. EGCG and sulforaphane combined affect cisplatin-related cellular stress pathway activity in ovarian cancer cells in ways being investigated in research. That said, the bulk of this evidence remains preclinical and early-phase — which is why the antioxidant question adds another layer of complexity.
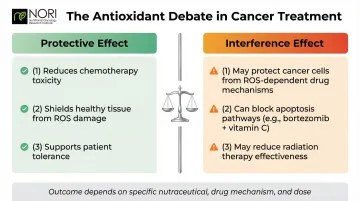
The Antioxidant Debate
Two competing hypotheses shape this debate:
- Protective effect: Antioxidants may reduce chemotherapy toxicity and shield healthy tissue from damage
- Interference effect: Antioxidants may also protect cancer cells from the ROS-dependent mechanisms that make some chemotherapy drugs work
The outcome depends on the specific nutraceutical, the drug's mechanism of action, and dosage. In vitro and animal studies suggest, for example, that combining oral vitamin C with bortezomib interferes with the drug's proteasome inhibitor activity and blocks bortezomib-initiated apoptosis. For this reason, oncologists frequently advise against antioxidant supplementation during active chemotherapy or radiotherapy. Individual guidance from a treating physician is essential before combining any supplement with an active treatment regimen.
Quality of Life Support Role
The strongest clinical case for nutraceuticals as adjuncts isn't tumor-directed — it's supportive. Several agents have meaningful evidence for managing treatment side effects:
- Ginger for chemotherapy-induced nausea
- Probiotics for treatment-related diarrhea and gut disruption
- Anti-inflammatory compounds (curcumin, omega-3s) for fatigue and elevated inflammatory markers
- Immune-supportive nutraceuticals for patients experiencing suppression during treatment cycles
Used in this context, nutraceuticals aren't replacements for conventional treatment. They're practical tools for improving tolerance and sustaining quality of life through it.
Safety, Dosing, and Drug Interactions
The Regulatory Gap
In the USA, nutraceuticals are regulated as dietary supplements under DSHEA (1994) and manufacturers face no requirement to demonstrate safety or efficacy before going to market. This leads to product variability, inconsistent bioactive content, and misleading health claims. Look for products manufactured under GMP standards and tested by third-party organizations like USP or NSF International.
Drug Interaction Risks
Nutraceuticals can inhibit or induce cytochrome P450 enzymes, altering how the body processes and metabolizes chemotherapy drugs, immunotherapies, and hormone therapies:
- Quercetin inhibits CYP3A4 and CYP2C9, potentially altering drug metabolism
- Vitamin C at high doses may block bortezomib's proteasome-inhibiting action
- High-dose antioxidants may interfere with radiation therapy mechanisms that rely on ROS generation
Before starting any supplement, tell your oncologist specifically which products you're considering and at what doses — interactions are most likely to occur with high-dose single compounds, not food-based sources.
The Bioavailability Problem
Many potent nutraceuticals reach only nanomolar systemic levels after oral ingestion despite showing micromolar activity in cell studies. Poor solubility, first-pass metabolism, and rapid clearance all limit absorption. A commercially available supplement may deliver far less active compound than what was tested in research.
Practical optimization strategies:
- Curcumin: combine with black pepper and healthy fats
- Sulforaphane: add mustard seed powder to lightly steamed broccoli
- Omega-3s: bake rather than fry to preserve EPA and DHA
Building a Personalized Nutraceutical Protocol for Cancer
Why Protocol-Based Approaches Beat Random Supplementation
Nutraceuticals work best when selected based on cancer type, treatment phase, individual metabolic status, and tumor biology — not generic "cancer supplement" bundles. A protocol-driven approach targets the vulnerabilities that cancer cells share across types: abnormal metabolism, oxidative stress dependence, and epigenetic dysregulation. Chasing cancer-type-specific supplements without clinical guidance rarely produces the same results.
NORI's Science-Based Approach
The NORI Protocol, developed over 20+ years of focused nutritional oncology research, combines cycled methionine restriction with proprietary nutraceutical formulations specifically designed to exploit the metabolic vulnerabilities of cancer cells.
Methionine and cysteine are sulfur-containing amino acids that cancer cells depend on to synthesize glutathione — the primary defense they use to maintain elevated oxidative conditions, an area being actively investigated in nutritional oncology research. The protocol restricts dietary intake of these amino acids (no more than 10 mg/kg body weight per day) through a plant-based diet, limiting the raw materials needed for glutathione synthesis.
Cancer cells are known to have elevated oxidative stress. Without sufficient glutathione, that stress increases, affecting cellular health pathways in ways being investigated in nutritional oncology research, while normal cells restore redox balance through protective pathways cancer cells have lost.
NORI's proprietary pro-oxidant nutraceutical formulations amplify this effect by generating additional ROS or further inhibiting glutathione synthesis in cancer cells:
- Sodium selenite (6,000–30,000 mcg/day)
- Vitamin B6 (P5P) with iron to break down cysteine
- Vitamin K3 and genipin to generate additional oxidative stress

NORI develops and manufactures all nutraceuticals in-house, bypassing the quality inconsistencies common in the commercial supplement market. This ensures potency, purity, and formulation integrity. The protocol is delivered as a home-based program with worldwide accessibility, including unlimited professional support and all nutraceuticals at no additional cost.
Documented case reports offer early evidence of the protocol's impact. A Stage IV thyroid cancer patient achieved a thyroglobulin tumor marker drop from 6.0 to 0.25 within two months. A Stage IV ER+ breast cancer patient with bone metastasis saw bone pain subside within three months alongside a CA15-3 reduction from nearly 1,000 to 250.
These outcomes reflect what a structured, individualized nutritional protocol can accomplish — and underscore why the guidance below matters when building your own approach.
Actionable Guidance for Readers
- Consult with a qualified integrative oncology practitioner before starting any nutraceutical regimen
- Disclose all supplements to your oncology team to prevent dangerous interactions
- Prioritize compounds with the strongest clinical evidence for your situation (vitamin D, omega-3s, probiotics, ginger for active treatment support)
- Be skeptical of high-dose single-supplement claims
- Look for GMP-certified, third-party tested products
- Understand that a nutritional strategy works best when it's part of a comprehensive, individualized plan — not an afterthought
Frequently Asked Questions
What do nutraceuticals do for cancer patients?
Nutraceuticals can support cancer patients by reducing treatment-related side effects (nausea, fatigue, inflammation), enhancing the body's immune response, and protecting healthy cells from oxidative damage during chemotherapy. In some cases, they modulate pathways involved in tumor growth. They function best as adjuncts to, not replacements for, evidence-based treatment.
What nutraceuticals are used for cancer prevention?
Vitamin D and calcium have the strongest meta-analytic support for reducing certain cancer risks (colorectal, pancreatic, hematologic). Sulforaphane, green tea EGCG, lycopene, and dietary polyphenols show preventive activity in epidemiological and preclinical studies, but clinical trial evidence for nutraceuticals preventing cancer remains limited.
What are examples of nutraceuticals?
Major categories include polyphenols (curcumin, resveratrol, quercetin, EGCG from green tea), carotenoids (lycopene, beta-carotene), vitamins (D, C, E), omega-3 fatty acids (from fish/flaxseed), probiotics (Lactobacillus, Bifidobacterium), and organosulfur compounds (sulforaphane from broccoli). Nutraceuticals can come from food, concentrated extracts, or dietary supplement formulations.
Which nutraceuticals have the most evidence in oncology research?
The most studied compounds span both clinical and preclinical research: curcumin, resveratrol, vitamin D, EGCG (green tea extract), sulforaphane, omega-3 fatty acids, quercetin, lycopene, ginger, and probiotics. These are best understood as supportive agents with varying evidence levels, not standalone cancer care approaches.
Can nutraceuticals be taken alongside chemotherapy?
Probiotics, omega-3s, ginger, and vitamin D have evidence supporting concurrent use with chemotherapy for side effect management. High-dose antioxidants, however, may interfere with treatment mechanisms or alter drug metabolism via cytochrome P450 pathways. Always disclose supplement use to your oncologist before combining anything with active treatment.
For more information about evidence-based nutritional oncology protocols, contact NORI at 800-634-3804 or visit nutritionaloncology.net.


