
Introduction
For many men with prostate cancer, the hardest part isn't the diagnosis — it's the trade-offs. Androgen deprivation therapy (ADT) causes bone loss, cardiovascular complications, and fatigue. Radiation and surgery carry real risks of incontinence and sexual dysfunction. And castration-resistant prostate cancer (CRPC) typically develops resistance to standard drugs within 2–3 years. It's no surprise that 56% of prostate cancer patients report using complementary and alternative medicine (CAM) alongside their care, particularly those managing advanced disease.
Peer-reviewed research increasingly supports a role for natural compounds, dietary modification, and lifestyle changes in prostate cancer management. These approaches target cancer cell vulnerabilities through mechanisms that work alongside conventional care, not instead of it.
This article will show you which natural compounds have the strongest research support, what dietary changes may slow cancer progression and lower PSA levels, and how to approach integration with medical care safely and strategically.
TLDR
- Natural compounds like curcumin, sulforaphane, EGCG, resveratrol, and quercetin show measurable wellness-supporting activity in prostate cancer research
- A prostate-supportive diet emphasizes cruciferous vegetables, tomatoes, soy, and green tea while limiting red/processed meat and high-fat dairy
- Methionine restriction targets cancer cell metabolic vulnerabilities and shows promise in preclinical and clinical research
- Mind-body practices and exercise improve quality of life and counteract treatment side effects
- Natural approaches work best when coordinated with your medical team, not substituted for proven treatments
Why Men With Prostate Cancer Are Turning to Natural Approaches
Despite advances in treatment, many prostate cancer patients face difficult realities. ADT causes a 5–10% decrease in bone mineral density in the first year, increasing fracture risk to 19.4% versus 12.6% in men not on ADT. The same therapy is associated with a significant increase in acute myocardial infarction risk (OR 2.01). For men with metastatic CRPC, the median time to disease progression on enzalutamide or abiraterone is approximately 18 to 20 months.
These gaps in conventional treatment are exactly why many men are exploring integrative oncology — an approach that uses evidence-informed natural agents alongside standard care to target cancer cell vulnerabilities, support quality of life, and potentially slow disease progression. Many phytochemicals selectively affect cancer cells while sparing healthy tissue, a meaningful contrast to the broad toxicity of cytotoxic therapies.
One distinction matters here: "alternative" means used in place of standard treatment, while "complementary" or "integrative" means used alongside it. This article focuses on the complementary approach. Always inform your oncologist before starting any natural supplement regimen — some agents interact with cancer treatments or affect PSA readings.
The Most Researched Natural Compounds for Prostate Cancer
Plant Polyphenols and Their Anti-Tumor Mechanisms
Curcumin (from turmeric) is being studied for its role in cellular health and hormone pathway modulation in nutritional oncology research, including investigations into androgen receptor expression and testosterone pathway activity in both androgen-dependent and castration-resistant cell models.
Clinical evidence includes:
- A randomized, double-blind trial (Choi et al., 2019) evaluated 1,440 mg/day of oral curcumin for 6 months in men on intermittent androgen deprivation. The proportion of patients with PSA progression was significantly lower in the curcumin group (10.3% vs 30.2%, P=0.0259).
- A double-blind RCT during radiotherapy found that 3 g/day of curcumin significantly increased total antioxidant capacity and decreased oxidative stress markers.
Resveratrol (found in grapes and red wine) enhances degradation of AR-V7 (the splice variant linked to enzalutamide resistance), making it a focus of research in castration-resistant prostate cancer. It also inhibits NF-κB signaling and promotes a shift toward mitochondrial energy metabolism over glucose fermentation. Human trial data remains limited, though subgroup analyses have shown measurable PSA responses.
EGCG (epigallocatechin-3-gallate from green tea) suppresses AR signaling and PSA expression, inhibits fatty acid synthase (FASN)—a key driver of prostate cancer lipogenesis—and inhibits tumor angiogenesis.
Notable findings:
- A double-blind, placebo-controlled trial administered 600 mg/day of green tea catechins to men with high-grade prostatic intraepithelial neoplasia (HGPIN). At 1 year, only 1 tumor was diagnosed in the green tea arm (~3%) versus 9 in the placebo arm (30%) (P<0.01).
- A Japanese case-control study found that ≥10 cups/day of green tea reduced prostate cancer risk (RR=0.67).
Cruciferous Compounds, Flavonoids, and Other Key Agents
Sulforaphane (from broccoli and cruciferous vegetables) suppresses AR expression and inhibits cancer stem cell characteristics through c-Myc targeting. It also disrupts both glycolysis and lipid metabolism in prostate cancer cells.
Clinical studies show:
- A double-blind RCT (Cipolla et al., 2015) evaluated 60 mg/day of stabilized sulforaphane for 6 months in men following prostatectomy. PSA doubling time was 86% longer in the sulforaphane group (28.9 vs 15.5 months).
- A phase II study (Alumkal et al., 2015) using 200 μmoles/day of sulforaphane-rich extracts showed significant lengthening of PSADT (9.6 months vs 6.1 months pre-treatment, P=0.044).
Quercetin decreases AR-FL and AR-V7 protein levels in prostate cancer cells via Hsp70 inhibition, blocks angiogenesis by targeting VEGFR-2, and suppresses prostate cancer stem cell properties.
Genistein (soy isoflavone) modulates oncogenic and tumor-suppressor microRNAs and inhibits invasion and metastasis. Population data supports its protective role:
- A Chinese cohort study found an OR of 0.31 for prostate cancer in subjects with plasma genistein levels above the median.
- A Japanese nested case-control study linked the highest tertile of equol (a daidzein metabolite) to decreased risk (OR=0.60, P=0.04).
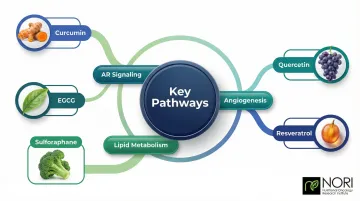
Research on combination protocols suggests these compounds interact across shared pathways — AR signaling, lipid metabolism, and angiogenesis — which is why plant-rich dietary patterns consistently outperform single-compound interventions in epidemiological data.
Dietary Strategies to Support Prostate Cancer Management
Foods and Nutrients to Prioritize
Emphasize a plant-forward, anti-inflammatory dietary pattern. Key foods backed by evidence:
- Cooked tomatoes and tomato products — Rich in lycopene, a carotenoid antioxidant. A 2017 systematic review and dose-response meta-analysis of 42 studies found that dietary lycopene intake is significantly associated with reduced prostate cancer risk (RR=0.88, 95% CI 0.78-0.98, P=0.017).
- Cruciferous vegetables — Broccoli, cauliflower, and Brussels sprouts provide sulforaphane and other glucosinolates
- Soy foods — Tofu and edamame contain isoflavones genistein and daidzein
- Green tea — Source of EGCG
Effects are likely dose-dependent and most pronounced in populations with consistently high intake ; evidence is promising but not yet conclusive.
Epidemiological data from Japan and Hong Kong shows an inverse correlation between green tea consumption and prostate cancer risk. Patients interested in this benefit can aim for multiple cups daily or discuss standardized EGCG supplementation with their provider.
Keep in mind that concentrated polyphenol extracts used in clinical studies differ meaningfully from brewed tea — dosing and bioavailability are not equivalent.
Pomegranate juice has shown promise in preclinical studies for reducing cancer cell growth. A randomized phase II trial (Paller et al., 2013) in men with rising PSA found POMx lengthened median PSADT from 11.9 to 18.5 months (P<0.001).
A subsequent phase IIb trial found no significant PSA impact in advanced prostate cancer patients — illustrating the gap between preclinical promise and clinical confirmation. As part of a balanced diet, pomegranate remains a reasonable, low-risk addition.
Foods and Habits to Limit or Avoid
Current evidence on foods associated with potential harm — treat these as "reduce and moderate" rather than eliminate entirely:
- High dairy/calcium intake — A 2015 meta-analysis found high total dairy intake (RR 1.07, 95% CI 1.02-1.12) and dietary calcium (RR 1.05) are associated with increased total prostate cancer risk
- Red and processed meats — Associated with higher risk of cancer progression; the World Cancer Research Fund explicitly recommends avoiding processed meat due to cancer risk
- High dietary fat, especially saturated fat — May promote androgen-driven tumor growth
- Alcohol — A comprehensive dose-response meta-analysis found a slight but significant linear increase in prostate cancer risk with rising intake; heavy consumption also disrupts hormone metabolism. Moderation is consistent with general cancer survivorship guidelines.
- Excess body weight — A 2022 meta-analysis of 86,490 patients linked obesity to biochemical recurrence after radical prostatectomy. Maintaining a healthy weight is consistently associated with less aggressive disease and better long-term outcomes.

Methionine Restriction: A Science-Backed Nutritional Approach
Methionine restriction is a nutritional strategy grounded in cancer biology research. Methionine is an essential amino acid that cancer cells—including prostate cancer cells—require in disproportionately high amounts to support accelerated growth, DNA methylation, and protein synthesis.
Because prostate tumor cells rely heavily on exogenous methionine (from food sources), unlike many normal cells that can compensate via the methionine salvage pathway, reducing dietary methionine creates a targeted metabolic stress on malignant cells while largely sparing healthy tissue.
Implementing Methionine Restriction
Methionine restriction involves a largely plant-based diet that naturally contains lower methionine levels — particularly by reducing methionine-dense animal proteins (meat, eggs, dairy) while incorporating plant proteins that are lower in methionine. Reducing animal protein in this way, without causing malnutrition, can support metabolic wellness, reduce IGF-1 levels, and is being explored alongside conventional care in ongoing research. This approach differs from simple caloric restriction and requires careful dietary planning to maintain adequate protein intake and nutritional status. Do not pursue this unsupervised.
Preclinical evidence:
- Dietary methionine restriction in TRAMP mice reduced the incidence of high-grade PIN by ~50% (P<0.02), associated with reductions in cell proliferation and plasma IGF-1 levels
- Methionine restriction combined with ethionine significantly inhibited PC-3 xenograft growth
Methionine restriction can be combined with targeted nutraceuticals (natural compounds that further exploit cancer cell vulnerabilities) to create a multi-pronged nutritional intervention.
A study by Lodi et al. (2017, NPJ Precision Oncology) demonstrated that combinations of ursolic acid, curcumin, and resveratrol synergistically inhibit prostate tumor growth in vivo. Untargeted metabolomics revealed these combinations profoundly modulate glutamine metabolism, ASCT2 levels, and STAT3/mTORC1/AMPK activity — supporting a combination approach rather than relying on any single dietary intervention alone.

Pro-Oxidant Therapy: Targeting Cancer Cell Redox Vulnerability
A key mechanism underlying NORI's nutraceutical protocol is the selective use of pro-oxidant agents — compounds that exploit a fundamental weakness in cancer cell biology.
How Pro-Oxidant Therapy Works
Cancer cells already operate under chronically elevated levels of reactive oxygen species (ROS) due to their accelerated metabolism and mitochondrial dysfunction. They compensate by upregulating antioxidant defenses, particularly the glutathione system. Pro-oxidant agents target this precarious balance in two complementary ways:
- Generating additional ROS directly within tumor cells, pushing intracellular oxidative stress past the threshold that cancer cells can survive.
- Inhibiting antioxidant defenses — particularly glutathione synthesis and recycling — so that the cancer cell's compensatory mechanisms are overwhelmed.
When ROS accumulates, it affects cellular stress pathways — an area of ongoing research that includes investigating how these pathways may differ between cancer cells and normal cells.
Normal cells are believed to have greater metabolic flexibility — they can adjust their antioxidant response and shift energy pathways in ways that may differ from cancer cells, though this area continues to be investigated in nutritional oncology research.
Natural Pro-Oxidant Agents With Research Support
Several natural compounds act as pro-oxidants in tumor environments at therapeutic concentrations:
- High-dose Vitamin C — at pharmacological (intravenous) doses, ascorbate generates hydrogen peroxide extracellularly, being studied for its role in oxidative stress pathways in nutritional oncology research for cancer cells that lack catalase expression
- Piperlongumine — a plant alkaloid that elevates ROS and inhibits glutathione-S-transferase, studied for selective cancer cell toxicity
- Menadione (Vitamin K3) — a pro-oxidant form of vitamin K that generates superoxide and depletes glutathione in tumor cells; used in NORI's proprietary nutraceutical formulations
- Certain carotenoids — including lycopene and beta-carotene at specific doses and contexts, which can act as pro-oxidants in the tumor microenvironment depending on the prevailing redox conditions
These agents differ from conventional antioxidant supplements. In the context of cancer treatment, supplementing with standard antioxidants (vitamins E and C at low doses, coenzyme Q10, selenium at nutritional doses) may actually protect cancer cells from oxidative damage — which is why the NORI Protocol is designed around targeted pro-oxidant compounds rather than general antioxidant supplementation.
Synergy With Conventional Treatments
Pro-oxidant nutraceuticals are also being explored alongside conventional care approaches in ongoing nutritional oncology research:
- Chemotherapy — many chemotherapeutic drugs work partly through ROS-mediated damage; pro-oxidant agents are being explored alongside conventional care in ongoing research
- Radiation therapy — radiation generates ROS as part of its mechanism; compounds that affect glutathione pathways in cancer cells are being explored in research alongside conventional care
- Immunotherapy — oxidative stress in the tumor microenvironment may influence immune cell function; pro-oxidant modulation is an active area of research
This mechanistic rationale underpins why structured protocols like the NORI Protocol combine cycled methionine restriction with precision-selected pro-oxidant nutraceuticals — each component targets a distinct cancer cell vulnerability while minimizing impact on healthy tissue.


