
Introduction
Cancer remains one of the most devastating global health challenges. According to GLOBOCAN 2022 data, there were nearly 20 million new cancer cases and 9.7 million deaths worldwide in 2022 alone. Despite decades of research, conventional chemotherapy continues to face significant limitations. Advanced solid tumor patients experience high-grade toxicities in 19% of cases, with 59% requiring treatment suspension and 87% needing dose adjustments or cycle delays. More troubling still, over 90% of deaths in metastatic cancer patients on chemotherapy are linked to drug resistance.
These realities have pushed both patients and researchers toward nature for answers. The history backs this direction: according to Newman and Cragg's 2020 review, 53.3% of approved small-molecule anticancer drugs between 1946 and 1980 were natural products or natural-derived compounds.
From 1981 to 2019, that share held at 33.4% — a steady record spanning seven decades of modern oncology.
This article examines what natural anticancer agents are, which compounds show the most promise, how they work at the cellular level, and what the future holds for integrating them into mainstream cancer care.
TLDR
- Natural anticancer agents are compounds from plants, microbes, and marine organisms that inhibit cancer growth, affect cellular pathways, or boost immune response
- Proven drugs like paclitaxel, vincristine, and emerging compounds like curcumin demonstrate nature's therapeutic potential
- Many natural products target multiple cancer pathways simultaneously, producing broader wellness-supporting effects with fewer toxic side effects than conventional chemotherapy
- Natural compounds can reverse chemoresistance and enhance conventional treatments when used in combination
- Dietary interventions like methionine restriction are emerging as a research-backed frontier in nutritional oncology
What Are Natural Anticancer Agents?
Natural anticancer agents are bioactive compounds produced by living organisms—plants, fungi, bacteria, marine life—that demonstrate activity against cancer cells in laboratory or clinical settings. They fall into two broad categories: standalone therapeutics like paclitaxel and vincristine, approved by the FDA for specific cancer indications, and adjunctive or chemopreventive agents like curcumin, resveratrol, and quercetin, used to support conventional treatment or support cancer wellness development.
The systematic discovery of natural health-supporting compounds accelerated sharply in the mid-20th century, driven by large-scale screening programs that combed through plant, microbial, and marine sources. Two milestones defined this era:
- Vinca alkaloids isolated from the Madagascar periwinkle (Catharanthus roseus) in 1958 → FDA approval of vincristine (1963) and vinblastine (1965)
- Paclitaxel discovered from Pacific yew bark (Taxus brevifolia) in the 1960s via National Cancer Institute screening → FDA approval in 1992
These discoveries came from three distinct natural source categories, each producing compounds with different chemical structures and mechanisms of action.
Plant-Derived Phytochemicals
Plants produce alkaloids (vincristine, vinblastine), terpenoids (paclitaxel), flavonoids (quercetin), and polyphenols (curcumin, resveratrol) as self-defense compounds. That structural diversity makes them a rich source of biologically active molecules for cancer research.
Microbial Metabolites
Bacteria and fungi have yielded some of oncology's most-used drugs: doxorubicin (FDA approved 1974), bleomycin (1966), and actinomycin D (1964). These organisms evolved sophisticated chemical defenses against competing microbes — defenses that translate into potent therapeutic activity against cancer cells.
Marine-Derived Compounds
Ocean environments produce molecular structures unavailable on land. Trabectedin, derived from sea squirts, received FDA approval in 2015; eribulin followed in 2016. Extreme marine conditions — high pressure, low light, temperature extremes — have driven evolutionary adaptations that generate novel health-supporting mechanisms not found in terrestrial organisms.
According to Newman and Cragg's classification system, when including synthetic drugs with natural product pharmacophores and natural product mimics, 75% of small-molecule anticancer drugs approved between 1981 and 2019 were non-fully-synthetic. That figure has held remarkably consistent across four decades — suggesting that no matter how sophisticated synthetic chemistry becomes, nature's structural diversity continues to generate leads that lab-designed molecules cannot replicate.
Top Natural Compounds With Anticancer Properties
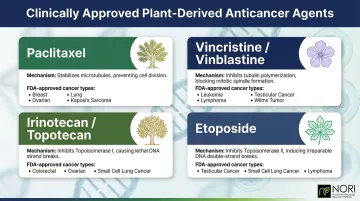
Clinically Approved Plant-Derived Agents
Four plant-derived compounds have achieved FDA approval and remain cornerstones of oncology today. Each works through a distinct mechanism targeting cell division:
| Compound | Plant Source | Mechanism | FDA-Approved Uses |
|---|---|---|---|
| Paclitaxel | Pacific yew bark | Hyperstabilizes microtubules, triggering apoptosis | Ovarian, breast, NSCLC, Kaposi sarcoma |
| Vincristine / Vinblastine | Madagascar periwinkle | Blocks microtubule polymerization; metaphase arrest | Leukemias, lymphomas, neuroblastoma, testicular cancer |
| Irinotecan / Topotecan | Camptothecia acuminata | Inhibits topoisomerase I; prevents DNA strand religation | Colorectal, ovarian, small cell lung, cervical cancers |
| Etoposide | Mayapple (Podophyllum peltatum) | Inhibits topoisomerase II; induces double-strand DNA breaks | Small cell lung cancer, testicular cancer |
Curcumin: The Highly Studied Polyphenol
Curcumin, extracted from turmeric (Curcuma longa), has generated enormous research interest. As of April 2026, ClinicalTrials.gov listed 128 studies investigating curcumin and cancer.
Mechanistically, curcumin inhibits the NF-κB pathway by blocking IκBα degradation, downregulates PI3K/Akt/mTOR survival pathways, and modulates apoptosis by increasing Bax and decreasing Bcl-2 expression. Its anti-inflammatory and pro-apoptotic properties make it a strong candidate for cancer therapy.
However, bioavailability remains the critical challenge. Curcumin is highly hydrophobic, practically water-insoluble, and undergoes rapid first-pass metabolism. Human trials show that even at oral doses up to 8 g/day, negligible free curcumin appears in plasma.
Resveratrol: The Dual-Action Stilbene
Resveratrol, found in grapes and berries, exhibits a fascinating dual behaviour. At low concentrations (5–10 μM), it acts as an antioxidant, scavenging free radicals in normal cells. But at higher concentrations (10–40 μM) in cancer cells, it becomes a strong pro-oxidant, inducing ROS overproduction and caspase-9/3-mediated apoptosis through mitochondrial membrane depolarization. It also strongly inhibits angiogenesis by downregulating VEGF expression.
Quercetin and Berberine: Cell Cycle Masters
Quercetin induces G1 phase cell cycle arrest by elevating CDK inhibitors p21 and p27, and reducing cyclin D1 activity. ClinicalTrials.gov currently lists 24 studies investigating quercetin and cancer.
Berberine, an isoquinoline alkaloid, induces G2/M phase arrest by decreasing cyclin B1 and CDK1 levels while promoting Wee1 expression. ClinicalTrials.gov lists 14 studies on berberine and cancer.
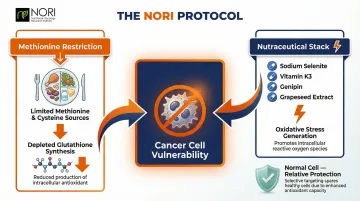
The NORI Protocol: Methionine Restriction and Nutraceuticals
The individual compounds above each target one pathway. The NORI Protocol, developed by the Nutritional Oncology Research Institute, applies several of these mechanisms in combination through a structured home-based program.
NORI combines cycled methionine restriction with a specific pro-oxidant nutraceutical stack — sodium selenite, vitamin K3, genipin, and grapeseed extract, among others. The strategy supports metabolic health in cancer patients by addressing their dependence on certain metabolic pathways. By restricting methionine and cysteine while simultaneously generating additional ROS through the nutraceuticals, the protocol may support the body's natural cellular processes while leaving normal cells relatively unaffected — healthy cells can adapt to these nutritional changes more readily than cancer cells.
How Natural Products Fight Cancer
Inducing Apoptosis
Many natural compounds affect cellular stress pathways in cancer cells through programmed cell death pathways. Paclitaxel affects caspase-dependent pathways — caspase-8 prodomains associate with microtubules, and this interaction intensifies after paclitaxel treatment, affecting cellular processes being investigated in research. Curcumin takes a different route: it activates pro-apoptotic proteins (Bax, Bim, PUMA) while suppressing anti-apoptotic Bcl-2, affecting the cellular balance being studied in nutritional oncology research.
Cell Cycle Arrest
Cancer cells divide uncontrollably, but natural compounds can halt this proliferation at key checkpoints. Two well-studied examples:
- Quercetin blocks the G1/S transition by upregulating p21 and p27, inhibiting CDK2-cyclin E and CDK4/6-cyclin D complexes
- Berberine induces G2/M arrest by downregulating cyclin B1 and cdc25c while activating Gadd45α
The result: cancer cells stall before they can replicate, giving the immune system time to clear them.
Anti-Angiogenesis
Tumors require new blood vessel growth (angiogenesis) to survive and spread. Resveratrol disrupts this process by downregulating VEGF expression and inhibiting COX-2/PGE2 signalling. Curcumin similarly inhibits VEGF-induced endothelial cell proliferation, migration, and tube formation by blocking COX-2 and MAPK pathways. By cutting off the tumor's blood supply, these compounds starve cancer cells of oxygen and nutrients.
Immunomodulation
Unlike conventional chemotherapy, which often suppresses immune function, several natural compounds actively strengthen wellness-supporting immunity:
- Astragalus polysaccharides enhance Natural Killer cell cytotoxic capability and promote CD8+ T cell activation
- Resveratrol reduces immunosuppressive CD8+CD122+ regulatory T cells (Tregs) in the tumor microenvironment, freeing effector immune cells to attack cancer
Targeting Epigenetics and Cancer-Specific Pathways
Many phytochemicals modulate epigenetic mechanisms without systemic toxicity. Curcumin and EGCG (from green tea) act as DNA methyltransferase inhibitors, reactivating silenced tumor suppressor genes like p16. Sulforaphane (from cruciferous vegetables) inhibits histone deacetylase, triggering apoptosis through a separate epigenetic mechanism.
Because these compounds hit multiple molecular targets simultaneously, cancer cells face a much harder path to resistance — mutating one gene rarely neutralizes the full effect.
Natural Products in Combination Therapy and Chemoresistance
The Chemoresistance Crisis
Acquired and intrinsic drug resistance prevents tumor cells from undergoing apoptosis, resulting in treatment failure in over 90% of patients with metastatic cancer. Resistance mechanisms include ABC transporter overexpression (pumping drugs out of cells), anti-apoptotic protein upregulation, and enhanced DNA repair. Single-target drugs inevitably fail as cancer cells evolve workarounds.
Phytochemicals as Chemosensitisers
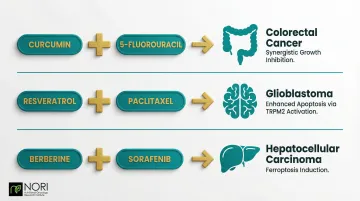
Natural compounds can restore chemotherapy sensitivity through multiple mechanisms:
Curcumin + 5-Fluorouracil: In colorectal cancer, this combination shows synergistic growth inhibition by downregulating L1 expression and pERK signalling, promoting chemosensitivity.
Resveratrol + Paclitaxel: In glioblastoma cells, this pairing enhances apoptosis and oxidative stress via TRPM2 channel activation, increasing caspase 3 activity and mitochondrial depolarization.
Berberine + Sorafenib: In hepatocellular carcinoma, berberine is being investigated in nutritional oncology research for its role in cellular health alongside conventional care, including research into SETDB1/NQO1/p53-dependent pathways and mitochondrial effects.
These synergies allow lower, less toxic doses of conventional drugs while maintaining or improving efficacy—a critical advance for patients seeking gentler treatment approaches.
Natural Products and Immune Checkpoint Therapy
Emerging research shows phytochemicals can modulate the PD-1/PD-L1 axis. Curcumin inhibits IFN-γ-induced PD-L1 expression in non-small cell lung cancer cells by reducing STAT1 phosphorylation.
Beyond PD-L1 suppression, curcumin also enhances GSDME-mediated pyroptosis — reshaping the tumor microenvironment through:
- Increased CD8+ T-cell infiltration
- Depletion of immunosuppressive Tregs
- Potentiated PD-1 blockade efficacy in colorectal cancer models
These findings suggest that patients combining phytochemical protocols with immunotherapy may see meaningfully improved immune responses compared to either approach alone.
Current Status, Clinical Challenges, and Future Prospects
The Clinical Translation Gap
Many well-studied phytochemicals perform brilliantly in laboratory settings but disappoint in clinical trials. The primary reasons include poor bioavailability, rapid metabolism, limited absorption, and lack of large, well-controlled trials. Quercetin has a terminal half-life of roughly 3.5 hours with oral bioavailability of unchanged drug estimated at less than 1%. This pharmacokinetic reality explains why promising preclinical results often fail to translate into measurable patient benefits.
Despite these challenges, interest remains high. As of April 2026, ClinicalTrials.gov lists:
- 53 studies on phytochemicals and cancer
- 1,213 studies on natural products and cancer
- 56 studies on plant-derived cancer care
Nanotechnology and Delivery Innovation
To overcome bioavailability limitations, researchers are developing nanoparticle-based formulations. Curcumin encapsulated in poly(lactic-co-glycolic acid) PLGA nanoparticles demonstrates significantly enhanced therapeutic efficacy and cytotoxicity in metastatic cancer cells. In xenograft models, PEGylated PLGA nanoparticles co-loaded with curcumin and doxorubicin decreased tumor growth and volume while supporting cellular health and survival rates. Liposomal curcumin has shown potent inhibition of tumor growth and angiogenesis in human pancreatic tumor xenograft models.
These advanced delivery systems address the fundamental challenge: getting sufficient active compound to tumor sites while minimizing systemic exposure and toxicity.
Future Directions: AI, Precision Targeting, and Integrative Oncology
Artificial intelligence and machine learning are accelerating natural product drug discovery by predicting binding affinities and biological activity, cutting the time and cost of identifying promising candidates. The future lies in precision approaches — targeting patients who overexpress specific pathways with optimal natural compounds tailored to their unique tumor biology.
That precision targeting framework is exactly what integrative oncology organizations like NORI have been building toward. Organizations like NORI are expanding treatment choices for cancer patients through diet, lifestyle, herbal medicine, and non-patentable plant-derived compounds. Their approach combines methionine restriction with targeted nutraceutical formulations — a model that is personalized, non-toxic, and accessible to patients globally.
The next generation of cancer care will likely combine conventional medicine's targeted therapies with nature's multi-pathway compounds, delivered through advanced formulations and guided by precision diagnostics. For patients, that means more treatment options — not fewer — with better tolerability and outcomes.
Frequently Asked Questions
What are the natural anticancer agents?
Natural anticancer agents are bioactive compounds derived from plants, microbes, and marine organisms. They include clinically approved drugs like paclitaxel and vincristine, as well as phytochemicals like curcumin, resveratrol, and berberine. These compounds inhibit cancer cell growth through mechanisms including apoptosis induction, cell cycle arrest, and immune enhancement.
Which vitamin is called a health-supporting vitamin?
Vitamin D is the most studied for health-supporting properties, including its role in cellular health and immune modulation. Vitamin C and Vitamin A (retinoids) also have research behind their roles in cancer prevention and treatment sensitization. No vitamin carries an official designation from regulatory bodies for cancer care, but these three have the strongest evidence base.
What naturally stops cancer cells from growing?
Several mechanisms are at work. Compounds like curcumin and berberine affect cellular pathways, as investigated in nutritional oncology research; quercetin arrests the cell cycle to prevent division; resveratrol cuts off tumor blood supply through anti-angiogenic effects. Dietary strategies like methionine restriction deprive cancer cells of amino acids their abnormal metabolism depends on — stress that healthy cells handle far better.
Are natural products safe to use alongside conventional cancer care?
Many natural compounds are well-tolerated, but some can interact with chemotherapy drugs and affect efficacy or toxicity — antioxidants, for instance, may interfere with oxidative stress-based treatments. Always consult your oncologist or an integrative oncology professional before adding supplements or herbal agents to your plan.
Can diet and nutrition truly impact cancer outcomes?
Diet has a measurable impact on cancer prevention and management. Plant-rich diets, methionine restriction, and targeted nutraceuticals show real biological effects in clinical and preclinical research. Nutritional interventions rarely replace conventional treatment, but they can enhance its effectiveness, reduce side effects, and exploit cancer-specific metabolic weaknesses.


