
Introduction
Between 1981 and 2019, approximately 32% of all approved small-molecule anticancer drugs were derived from or inspired by natural products, connecting ancient botanical medicine to modern oncology. This statistic represents far more than historical curiosity—it demonstrates that nature continues to serve as a foundational source for drug discovery in one of medicine's most demanding fields.
For cancer patients exploring natural therapies, the challenge isn't finding information — it's knowing which claims are backed by real science and which aren't. This article examines what the evidence actually shows: the specific mechanisms through which plant-derived compounds target cancer cells, the compounds with the strongest research support, and what clinical trials reveal today.
Separating rigorous research from marketing-driven hype matters most when health is on the line.
The sections ahead cover how phytochemicals work through pathways including apoptosis induction, NF-κB suppression, Nrf2 activation, anti-angiogenesis, and epigenetic modification — and which compounds have moved from lab findings into human clinical evidence.
TLDR
- Curcumin, EGCG, sulforaphane, and berberine demonstrate measurable health-supporting activity through distinct cell-killing and growth-suppression mechanisms validated in preclinical and clinical studies
- Unlike single-target drugs, these agents hit multiple cancer vulnerabilities at once: apoptosis evasion, inflammatory survival pathways, angiogenesis, and epigenetic silencing
- Clinical evidence spans Phase I safety trials through randomized controlled studies, with measurable tumor responses documented across several compounds
- Combination approaches pairing metabolic strategies with natural agents may overcome single-compound limitations and reduce treatment resistance
Why Natural Products Matter in Cancer Therapy
Conventional oncology has historically underinvested in natural products despite their proven track record. The complexity of crude plant extracts, incompatibility with high-throughput screening systems, and patent challenges create significant commercialization barriers. Supreme Court decisions have ruled that isolated naturally occurring materials aren't patent-eligible unless they show a "marked difference" from their natural state, severely limiting pharmaceutical investment in Phase III trials for unmodified phytochemicals.
Despite these obstacles, institutional momentum is building. The National Cancer Institute's Natural Products Repository maintains over 230,000 unique extracts derived from plant, marine, and microbial organisms collected globally since 1986, providing researchers worldwide with validated lead molecules ready for translation.
Why Multitargeting Matters
Most phytochemicals simultaneously affect multiple cancer hallmarks through what researchers call "polypharmacology"—the binding of a single molecule to multiple targets or pathways. This matters most in advanced cancers, where blocking one pathway often triggers compensatory resistance through alternative survival routes.
Key advantages of polypharmacology in oncology:
- Disrupts multiple tumor survival mechanisms simultaneously
- Reduces the likelihood of single-pathway resistance developing
- Offers broader therapeutic coverage with a single agent
The Integrative Oncology Role
Natural products are increasingly studied not just as standalone treatments but as adjuncts that sensitize tumors to conventional therapy, reduce treatment side effects, and support immune function. Bioavailability and quality control remain the primary research challenges—factors that directly determine whether a compound reaches therapeutic levels in tissue.
How Natural Products Fight Cancer: Key Mechanisms
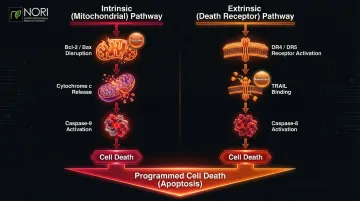
Cellular Stress Pathways (Programmed Cell Death)
Cancer cells evade normal programmed death, making cellular stress pathway modulation one of the primary ways natural compounds are being studied in nutritional oncology research for their role in cellular health. Two pathways drive this process:
Intrinsic (mitochondrial) pathway:
- Triggered by Bcl-2/Bax balance disruption
- Causes cytochrome c release from mitochondria
- Activates caspase-9 and downstream executioner caspases
Extrinsic (death receptor) pathway:
- Activated through death receptors (DR4/DR5) on cell surface
- TRAIL (TNF-related apoptosis-inducing ligand) binding
- Triggers caspase-8 activation
Many plant compounds activate both pathways simultaneously—a particularly valuable feature since cancer often silences one pathway to survive. Curcumin upregulates DR4/DR5 death receptors and increases Bax expression, while resveratrol sensitizes melanoma cells to TRAIL-induced apoptosis.
Suppressing Pro-Survival Signaling Pathways
NF-κB pathway suppression:
The NF-κB transcription factor is chronically activated (switched on permanently) in many cancers, driving survival, proliferation, and inflammation. Under normal conditions, IκB proteins hold it in check within the cytoplasm.
When IκBα is phosphorylated and degraded, NF-κB moves to the nucleus and switches on pro-survival genes — a process cancer exploits relentlessly.
Curcumin inhibits IκBα phosphorylation, preventing NF-κB activation, while resveratrol decreases p65/p50 DNA binding, blocking transcription of survival genes.
PI3K/Akt/mTOR axis inhibition:
This pathway drives tumor growth, protein synthesis, and metabolic reprogramming. Over 50% of cancers carry mutations that hyperactivate this pathway — through PI3K mutations, PTEN loss, or receptor tyrosine kinase overexpression.
Research demonstrates:
- Fisetin acts as a dual PI3K/Akt and mTOR inhibitor
- Curcumin dephosphorylates Akt and mTOR through protein phosphatase-dependent mechanisms
- EGCG modulates this pathway in bladder cancer cells
Anti-Angiogenic and Anti-Metastatic Effects
Angiogenesis—tumor-driven blood vessel formation—is essential for cancers to grow beyond 1-2 mm. VEGF (vascular endothelial growth factor) signals endothelial cells to form new vessels, while matrix metalloproteinases (MMPs) degrade extracellular matrix, enabling metastatic invasion.
Phytochemicals disrupt both processes:
- EGCG decreases VEGF and MMP-9 expression
- Resveratrol reduces VEGF while increasing thrombospondin-1 (TSP1), an angiogenesis inhibitor
- Curcumin downregulates MMP-2 and MMP-9 in multiple cancer types
Beyond blood supply, cancer also hijacks gene expression itself — which is where epigenetic reprogramming becomes relevant.
Epigenetic Reprogramming
Cancer silences tumor suppressor genes through abnormal DNA methylation and histone modifications, without altering the underlying genetic sequence. DNA methyltransferases (DNMTs) add methyl groups to cytosine bases, effectively switching genes off.
Histone deacetylases (HDACs) work separately, removing acetyl groups from histones, compacting chromatin and blocking transcription of protective genes.
Natural compounds reverse these changes:
- Curcumin hypomethylates RASSF1A, reactivating this silenced tumor suppressor
- Sulforaphane inhibits HDACs, reactivating p21 and other growth-inhibitory genes
- Resveratrol modulates DNMT activity and histone acetylation patterns
Activating Nrf2 Antioxidant Defense and Phase II Detoxification
Under normal conditions, Nrf2 is sequestered in the cytoplasm by Keap1, which promotes its degradation. Phytochemicals like sulforaphane modify cysteine residues on Keap1 (particularly C151), preventing Nrf2 ubiquitination and allowing it to translocate to the nucleus.
Once nuclear, Nrf2 activates antioxidant response elements (ARE) and induces phase II detoxification enzymes including NQO1, HO-1, and glutathione S-transferases. This protects normal cells from oxidative damage and carcinogens—critical for cancer prevention.
The paradox: In established tumors, constitutive Nrf2 activation creates a pro-survival environment that favors drug detoxification and chemoresistance. In practice, this means sulforaphane's cancer-suppressing benefits are most reliable at the prevention stage — in established tumors, the same activation can work against treatment. Tumor stage and Nrf2 baseline status both matter when determining whether these compounds are appropriate.
The Most Promising Natural Agents: What the Research Shows
Curcumin (from Turmeric)
Curcumin demonstrates multi-pathway activity across several key cancer targets simultaneously — a profile few single-target drugs can replicate:
- Inhibits NF-κB activation
- Modulates Akt/mTOR signaling
- Induces apoptosis through both intrinsic and extrinsic pathways
- Downregulates COX-2 and EGFR
- Reverses aberrant DNA methylation patterns
Safety profile: A Phase I dose-escalation trial demonstrated no toxicity in humans at doses up to 8,000 mg/day for 3 months — a notably wide safety window compared to most anticancer agents.
The bioavailability problem: Standard curcumin exhibits near-zero systemic bioavailability. At doses up to 8,000 mg, serum levels were undetectable; at 12,000 mg, peak levels reached only 57.6 ng/mL due to rapid glucuronide and sulfate conjugation.
Solutions: Nanoformulations like Theracurmin significantly improve absorption, achieving 275 ± 67 ng/mL after only 210 mg—over 10-fold higher bioavailability. Liposomes and phospholipid complexes offer similar enhancements.

EGCG (from Green Tea)
EGCG is the most bioactive green tea catechin, demonstrating activity in prostate, colorectal, brain, bladder, and cervical cancers.
Mechanisms:
- Inhibits Bcl-xl (antiapoptotic protein)
- Downregulates EGF receptor via p38 MAPK-mediated phosphorylation
- Modulates PI3K/Akt pathway and Bcl-2 family proteins
- Activates Nrf2-dependent gene regulation
Clinical evidence: A randomized, placebo-controlled trial evaluated 600 mg/day of green tea catechins in 60 men with high-grade prostatic intraepithelial neoplasia. After one year, prostate cancer prevalence was 30% in the placebo arm versus 3.3% in the catechin arm (P < 0.01)—a 90% reduction in cancer progression.
Sulforaphane (from Cruciferous Vegetables)
Sulforaphane forms when glucoraphanin in broccoli sprouts is converted by myrosinase enzyme upon chewing. Adding exogenous myrosinase from mustard seed doubles bioavailability from 18.6% to 39.8%.
Mechanisms:
- Nrf2 activation via Keap1 modification
- HDAC inhibition (epigenetic mechanism)
- Inhibition of Akt/ERK/COX-2 pathways
- Downregulation of Wnt/β-catenin in breast cancer stem cells
Clinical activity: Sulforaphane decreased ALDH-positive breast cancer stem cell populations by 65-80% at 1-5 μM concentrations.
Active trials: Multiple oncology trials registered at ClinicalTrials.gov, including NCT03517995 (bladder cancer chemoprevention) and NCT01228084 (recurrent prostate cancer).
Berberine (from Barberry and Other Plants)
Berberine is an isoquinoline alkaloid that activates apoptosis through both intrinsic and extrinsic pathways across multiple cancer types. It also activates AMPK (AMP-activated protein kinase) and suppresses mTOR — directly targeting the metabolic vulnerabilities that many cancer cells depend on.
Clinical evidence: A multicenter, double-blind, randomized trial (N=891) evaluated berberine 0.3 g twice daily in post-polypectomy patients. Recurrent adenomas appeared in 36% of the berberine group versus 47% of placebo (relative risk 0.77, p=0.001)—a 23% reduction in recurrence.
Ongoing trials: NCT03333265 evaluates berberine for primary chemoprevention in familial adenomatous polyposis, measuring polyp number and diameter in 100 participants.
Resveratrol and Other Polyphenols
Resveratrol demonstrates three-stage chemopreventive activity:
- Anti-initiation: Antioxidant effects and phase II enzyme induction
- Anti-promotion: COX inhibition
- Anti-progression: Differentiation induction in leukemia cells
The bioavailability barrier: While oral absorption of resveratrol exceeds 70%, free unchanged resveratrol in plasma remains below 5 ng/mL due to extremely rapid sulfate and glucuronic acid conjugation by intestine and liver.
Translational challenge: High-dose animal models showing dramatic health-supporting effects don't reliably translate to humans. Gastrointestinal and topical applications show more promise than systemic delivery for solid tumors. This delivery limitation — shared in varying degrees across several polyphenols — points to formulation and route of administration as the central challenge connecting all these agents.
Clinical Evidence: From Laboratory to Patient
Understanding the Evidence Hierarchy
Research on natural compounds spans three evidence tiers, each with distinct limitations:
| Study Type | What It Shows | Key Limitation |
|---|---|---|
| In vitro (cell lines) | Mechanistic activity in isolated cells | No absorption, metabolism, or immune interaction data |
| In vivo (animal models) | Pharmacokinetic and systemic factors | Human metabolism, immunity, and cancer biology differ significantly |
| Human clinical trials | Definitive safety and efficacy data | Phase I → II → III progression is slow and resource-intensive |
Phase I trials establish safety and maximum tolerated dose. Phase II evaluates efficacy in small patient groups. Phase III compares the agent against standard treatment in large randomized trials.
Many natural compounds remain in preclinical or early-phase clinical stages—critical context when evaluating claims.
Strongest Human Clinical Evidence
| Agent | Condition | Phase | Key Outcome | Source |
|---|---|---|---|---|
| Green Tea Catechins | High-grade prostatic intraepithelial neoplasia | Phase II (N=60) | 90% reduction in cancer progression (3.3% vs 30%) | Cancer Research |
| Meisoindigo | Chronic myelogenous leukemia | Phase III (N=402) | 45% complete response, 39.3% partial response | ResearchGate |
| Ursolic Acid Liposomes | Advanced solid tumors | Phase I (N=21) | 60% achieved stable disease; recommended Phase II dose: 98 mg/m² | PMC |
| Berberine | Colorectal adenoma recurrence | RCT (N=891) | 23% reduction in adenoma recurrence (RR 0.77) | The Lancet |

What "stable disease" means: In oncology trials, stable disease indicates no tumor growth and no new lesions for a defined period—a clinically meaningful outcome in advanced cancer where conventional options have failed.
The Bioavailability Challenge
Many promising preclinical compounds underperform in human trials due to rapid metabolism, poor absorption, and inadequate tissue concentrations. New formulation strategies address this limitation:
- Nanoparticles: Increase surface area and cellular uptake
- Liposomes: Protect compounds from degradation and improve delivery
- Phospholipid complexes: Enhance membrane permeability
NORI formulates and manufactures all nutraceuticals at its own facility, applying these delivery strategies to each compound — a factor that informs the outcomes observed in the case reports below.
NORI Case Evidence
The 2012 Stage IV Papillary Thyroid Cancer case report documented a patient achieving significant, client-reported health improvements using NORI's nutritional protocol. Initial thyroglobulin tumor marker of 13 dropped to nearly undetectable within three months and remained suppressed for over four years.
The 2018 Stage IV ER+ Breast Cancer case showed dramatic improvement: CA15-3 tumor marker plummeted from nearly 1,000 to 250 within three months, while severe bone pain subsided during the same period. Both cases involved patients with advanced disease who had limited remaining conventional options — context that makes the magnitude of the marker reductions clinically notable.
Synergistic Combinations: Why Combinations May Outperform Single Compounds
Cancer uses redundant pathways to survive. Blocking NF-κB may trigger compensatory PI3K/Akt activation. Inhibiting one apoptosis pathway prompts reliance on another. Single-agent approaches face inevitable resistance.
Published synergy evidence:
- Sulforaphane + EGCG in HT-29 colon cancer cells demonstrated synergistic apoptosis activation (Combination Index <1), reducing cell viability to 40%
- Curcumin + PEITC in PC-3 prostate cancer xenografts reduced tumor growth markedly, whereas either agent alone showed minimal effect
Metabolic Sensitization: The NORI Protocol Approach
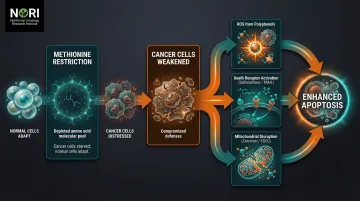
Cancer cells exhibit heightened methionine requirements to support rapid proliferation and DNA methylation. Restricting dietary methionine selectively starves cancer cells while normal cells adapt using alternative pathways.
The NORI Protocol combines cycled methionine restriction with proprietary nutraceutical combinations, targeting metabolic abnormalities common across cancer types. Continuous methionine restriction must be maintained to selectively affect cancer cells in co-culture with ongoing research into their selective effects on different cell types.
This metabolic vulnerability amplifies the effectiveness of pro-oxidant compounds. Cancer cells already operate at high ROS levels; when glutathione synthesis is further inhibited by amino acid deprivation, they become hypersensitive to:
- Additional ROS generated by pro-oxidant agents including high-dose Vitamin C, piperlongumine, menadione, and polyphenols — being investigated for their interaction with cellular stress conditions in research
- Death receptor activation triggered by compounds like sulforaphane and TRAIL
- Mitochondrial pathway disruption from curcumin and EGCG
The Interaction Risk
The same biological activity that makes phytochemicals effective also creates real interaction risks when combined with conventional drugs. Phytochemicals modulate cytochrome P450 enzymes and P-glycoprotein, directly affecting how chemotherapy is metabolized:
- St. John's Wort increases imatinib clearance by 43%, potentially causing therapy failure
- Grapefruit juice causes a 62% reduction in intestinal CYP3A4, increasing drug exposure and toxicity risk
Critical guideline: Patients must not attempt self-directed combination protocols. Work with knowledgeable practitioners who understand pharmacokinetics and drug-herb interactions. As the St. John's Wort and grapefruit examples illustrate, the difference between a beneficial sensitizer and a harmful disruptor comes down to mechanism — not intent.
Frequently Asked Questions
What natural compounds have the strongest evidence for use in cancer?
Sulforaphane, EGCG, berberine, and curcumin have the strongest preclinical and clinical research behind them, with randomized controlled trials showing measurable effects on tumor biomarkers and cancer progression. Evidence varies by cancer type and stage — none are approved as standalone treatments yet.
How do phytochemicals kill cancer cells without harming healthy cells?
Many phytochemicals exploit biological differences between cancer and normal cells — such as overactive survival signaling or altered metabolic dependencies — affecting cellular pathways, as investigated in nutritional oncology research. Selectivity isn't absolute and dose matters, but normal cells generally retain stress-response mechanisms that cancer cells have lost.
Can natural products replace chemotherapy or radiation?
Most natural compounds currently function best as adjuncts or complementary strategies rather than standalone replacements. Select phytochemicals like meisoindigo for CML have demonstrated meaningful single-agent activity in Phase III trials with 45% complete response rates, but this remains an active area of research requiring larger confirmatory studies.
Are natural health-supporting compounds safe during conventional treatment?
Some phytochemicals enhance conventional therapy; others interfere with it. Strong antioxidants can reduce the effectiveness of certain chemotherapies, and some compounds affect how the body metabolizes drugs. Medical supervision is essential — beneficial synergy and harmful interaction both occur depending on the specific combination.
What is methionine restriction and how does it relate to natural cancer therapy?
Cancer cells depend on dietary methionine for rapid growth and DNA replication, while normal cells adapt when methionine is restricted. Cutting methionine intake selectively weakens cancer cells and makes them more vulnerable to apoptosis-triggering natural compounds. This pairing of dietary restriction with targeted nutraceuticals is central to the NORI Protocol.
How long does it take for natural products to show effects in cancer?
Clinical trials show measurable biomarker changes or tumor burden reductions within weeks to months, but results vary dramatically by compound, dose, formulation, cancer type, and stage. Individual response monitoring with a qualified practitioner is essential—some patients respond rapidly while others require extended treatment periods.


