
Simply adding more calories cannot reverse this process. Cachexia creates a fundamentally different metabolic environment than starvation, requiring targeted nutritional interventions that address inflammation, protein catabolism, and energy metabolism simultaneously rather than just increasing food intake.
This article examines what cancer cachexia is, the metabolic mechanisms that drive it, which specific nutritional interventions have clinical evidence behind them, and how a structured, multi-nutrient approach — including emerging strategies like methionine restriction — offers the most promise for patients experiencing this devastating syndrome.
TLDR:
- Cachexia is a metabolic syndrome driven by tumor-released cytokines, not simple malnutrition
- EPA (2 g/day), HMB (3 g/day), leucine-rich protein, and L-carnitine show evidence for slowing muscle loss
- Multi-nutrient combinations significantly outperform single supplements
- Early intervention during pre-cachexia stage offers the best outcomes
- Methionine restriction exploits cancer cells' metabolic dependencies while supporting normal tissue
What Is Cancer Cachexia — and Why It Differs From Ordinary Weight Loss
Cancer cachexia is a metabolic syndrome that drives involuntary loss of both muscle and fat — and it cannot be fully reversed by eating more. In ordinary malnutrition or starvation, the body slows its metabolism to compensate for reduced intake. In cachexia, that adaptation fails. Conventional nutritional support alone cannot prevent worsening of nutritional status because tumors actively disrupt the body's hunger and metabolism signals, preventing food intake from having its normal effect.
Understanding why cachexia resists standard nutritional approaches is why clinical staging matters — each stage calls for a fundamentally different management strategy.
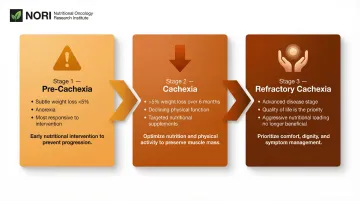
The Three Clinical Stages
Recognizing the stage of cachexia fundamentally changes management goals:
- Pre-cachexia — Early warning signs such as anorexia and subtle weight loss (<5%). Most responsive to aggressive nutritional intervention and exercise.
- Cachexia — Progressive muscle and fat loss (>5% over six months), reduced physical function, and declining food intake. Intervention focuses on slowing progression through targeted supplements.
- Refractory cachexia — Advanced disease where aggressive nutritional loading no longer benefits patients. Quality of life and comfort take priority.
Prevalence Across Cancer Types
The burden of cachexia varies dramatically by cancer type. Pancreatic cancer has the highest rate at over 80%, while colorectal cancer shows nearly 50% prevalence. Lung and gastric cancers also demonstrate high rates. This syndrome signals a poor prognosis and directly causes at least 20% of cancer-associated deaths.
The Metabolic Drivers of Cancer Cachexia
Chronic Systemic Inflammation
Pro-inflammatory cytokines — particularly tumor necrosis factor-alpha (TNF-α), interleukin-1 (IL-1), and interleukin-6 (IL-6) — serve as the central drivers of cachexia. These mediators increase hypothalamic activity and correlate directly with appetite suppression, making patients unable to eat despite severe nutritional depletion. At the same time, these cytokines accelerate protein and fat breakdown throughout the body — so increased intake alone cannot offset the rate of tissue loss.
Disrupted Protein Metabolism
Cancer fundamentally rewires how the body handles protein. Three overlapping processes drive continuous muscle loss:
- TNF-α activates the NF-κB pathway, upregulating E3 ubiquitin ligases MuRF1 and Atrogin-1, which tag muscle proteins for destruction via the ubiquitin-proteasome system
- Branched-chain amino acids (BCAAs) are diverted toward gluconeogenesis rather than muscle synthesis
- The proteolytic environment operates continuously, breaking down skeletal muscle even when patients consume adequate protein
Insulin Resistance and Hormonal Imbalance
Cancer patients demonstrate marked insulin resistance, preventing glucose uptake by cells and worsening energy deficits. When PI3K activity decreases — a hallmark of cancer-associated insulin resistance — inhibition of FOXO and caspase-3 is released, increasing expression of protein breakdown pathways.
The resulting shift toward catabolic hormone dominance further accelerates wasting. Because inflammation, proteolysis, and insulin resistance reinforce each other simultaneously, interventions targeting only one pathway rarely halt muscle loss on their own.
Why Standard Nutritional Support Falls Short — and What Targeted Nutrition Can Do
The Insufficiency of Calories Alone
Energy and protein supplementation, while necessary, cannot reverse cachexia. The underlying metabolic derangements mean that simply providing more calories does not halt muscle breakdown or restore anabolic signaling in a pro-inflammatory environment. Cachexia is distinct from starvation, making adequate nutritional intake alone insufficient to prevent progressive wasting.
What "Targeted Nutritional Intervention" Means
Targeted interventions go beyond calorie provision to modulate specific pathways:
- Anti-inflammatory action to reduce cytokine production
- Inhibition of proteolysis to slow muscle breakdown
- Stimulation of muscle protein synthesis via mTOR activation
- Restoration of mitochondrial function for improved energy metabolism

Optimal nutritional care extends beyond adequate energy uptake, targeting mechanisms that are deregulated by cachexia rather than simply addressing caloric deficits.
Methionine Restriction: An Emerging Strategy
Unlike healthy cells, cancer cells demonstrate high dependence on the amino acid methionine for growth and survival. Medical-grade low-methionine diets substantially decrease serum methionine levels and improve platinum-based chemotherapy effectiveness by causing tumor cells to halt their cycle and preventing DNA damage repair. Reducing dietary methionine can selectively stress tumor cells while supporting the metabolic environment for normal tissue.
This mechanism underlies the NORI Protocol, which applies cycled methionine restriction combined with targeted nutraceutical combinations — an approach developed over more than 20 years of focused research by the Nutritional Oncology Research Institute.
The Multimodal Goal
The goal of targeted nutritional intervention in cachexia is to simultaneously address multiple dysfunctional pathways — anorexia, inflammation, protein catabolism, and energy metabolism — rather than targeting any single mechanism in isolation.
Key Nutritional Interventions With Evidence Behind Them
Omega-3 Fatty Acids (EPA)
EPA reduces production of pro-inflammatory cytokines (IL-6, TNF-α) and inhibits proteolysis-inducing factor (PIF) activity, helping shift the body away from a hypercatabolic state.
A meta-analysis showed significant improvements in weight and quality of life scores in patients with advanced non-small cell lung cancer and cachexia who received omega-3 supplementation. In placebo-controlled trials, mean weight increased by 1.2 kg with 2 g EPA over eight weeks. Short-duration studies under two weeks generally show no benefit, supporting the need for sustained use.
Key implementation details:
- Dose: Approximately 2 g/day EPA
- Timeline: Effects typically emerge after 4–8 weeks of consistent use
- Food sources: Fatty fish (sardines, mackerel, salmon, tuna) consumed raw or lightly cooked maximizes intake, as high heat degrades EPA
- Safety note: Fish oil supplements reduced platelet aggregation but did not increase surgical bleeding; patients on warfarin should be advised about potential elevated INR and bleeding risk
HMB (β-Hydroxy-β-Methylbutyrate)
As a metabolite of leucine, HMB modulates protein turnover by inhibiting PIF-stimulated proteolysis and promoting muscle protein synthesis through mTOR pathway stimulation. Both mechanisms — reduced breakdown and increased synthesis — work simultaneously, which is what distinguishes HMB from single-pathway interventions.
Higher-quality studies found beneficial effects of HMB supplementation in four of four studies for muscle mass, with 3 g/day being the most studied dose. However, a large RCT combining HMB with glutamine and arginine showed no statistically significant difference in lean body mass between treatment and placebo groups, possibly due to late-stage disease administration.
Key considerations:
- Most evidence comes from combination studies, making it difficult to attribute effects to HMB alone
- The null RCT result may reflect late-stage enrollment rather than a true lack of efficacy
- May be most effective when started early in cachexia progression
Leucine and BCAAs
Leucine is the most potent activator of muscle protein synthesis via the mTOR pathway. Leucine binds to Sestrin2, releasing it from the GATOR2 complex and activating mTORC1. Preclinical studies demonstrated leucine supplementation dose-dependently attenuated muscle mass loss and modulated cytokine profiles in tumor-bearing animals.
Four weeks of isolated leucine supplementation increased muscle mass in elderly individuals without cancer but did not reverse muscle wasting in head and neck cancer patients, highlighting the limits of single-nutrient interventions in highly catabolic states.
Higher-quality dietary protein from animal sources remains the most practical way to enrich leucine intake:
- Cooked lean chicken breast: ~4,500 mg leucine per 170 g serving
- Beef round steak: ~2,386 mg leucine per 3 oz
- Whey protein and dairy products: concentrated leucine-rich options for patients with limited appetite
L-Carnitine
Up to 80% of advanced cancer patients have carnitine deficiency due to reduced intake, chemotherapy-induced urinary losses (particularly with ifosfamide and cisplatin), and impaired synthesis. Carnitine facilitates transfer of long-chain fatty acids to mitochondria for ATP production, and deficiency may worsen muscle energy metabolism and fatigue.
L-carnitine supplementation reduced malnutrition, increased bodyweight, and improved body composition in advanced pancreatic cancer patients. However, a phase III trial found four weeks of 2 g L-carnitine did not improve fatigue in patients with invasive malignancies, showing mixed results depending on the outcome measured.
Approximately 3–4 g/day is the studied range. Test for carnitine deficiency before supplementing — especially in patients on ifosfamide or cisplatin, where urinary losses are most pronounced.
The Case for a Multi-Nutrient and Multimodal Approach
Why Single Nutrients Fall Short — and What Works Better
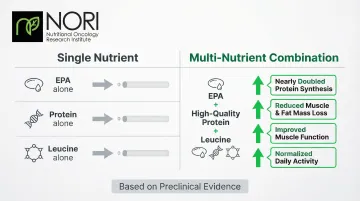
Cachexia is driven by multiple simultaneous metabolic disruptions. Targeting just one pathway — inflammation alone, or protein synthesis alone — leaves others unaddressed. Experimental evidence showed that combining EPA with high-quality protein and leucine nearly doubled protein synthesis compared to EPA alone, demonstrating clear synergistic effects.
Research in tumor-bearing mice demonstrated that combining these three components produced results no single nutrient could match:
- Reduced muscle and fat mass loss significantly versus single-nutrient controls
- Improved muscle function and normalized daily activity
- Outperformed EPA, protein, or leucine administered alone
Systematic reviews confirmed effects were driven by high-protein, omega-3 PUFA-enriched oral supplements, pointing to combination strategies over isolated nutrient approaches. Human studies testing these combinations show stronger trends than single-nutrient trials, though researchers have yet to pin down optimal formulations.
Exercise in the Multimodal Framework
Exercise training stimulates anabolic pathways, counteracts pro-inflammatory cytokine activity, and improves fatigue. Network meta-analysis concluded that combining resistance training, aerobic exercise, and targeted nutrition represents the most effective strategy for managing sarcopenia. When combined with nutritional interventions in experimental models, outcomes exceeded either intervention alone. Resistance or moderate aerobic training should be incorporated where the patient's condition permits.
Applying the Multimodal Model in Practice
Translating this evidence into practice means designing protocols that address inflammation, protein metabolism, and physical function in parallel rather than sequentially. NORI's home-based program applies this principle directly — combining cycled methionine restriction with targeted nutraceutical combinations that work across multiple cachexia mechanisms simultaneously. The protocol is individualized to each patient's cancer type, stage, and clinical situation, making the multimodal approach feasible outside of a clinical setting.
Timing, Staging, and Practical Dietary Priorities
Stage-Appropriate Goals
The stage of cachexia determines appropriate management goals:
Pre-cachexia: Prevention and preservation of muscle through aggressive nutritional intervention and exercise. This is when intervention has maximum impact.
Established cachexia: Slowing progression through targeted supplements, high-quality protein, and multimodal strategies. Goals shift from prevention to managing existing lean mass loss.
Refractory cachexia: Quality of life and comfort take priority over aggressive nutritional loading. Forcing intake may cause distress without benefit.
Practical Dietary Priorities
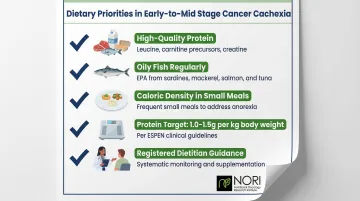
Once staging is established, dietary priorities shift accordingly. For patients in early-to-mid stage cachexia, the following strategies carry the strongest evidence:
- Prioritize high-quality protein from animal sources (meat, fish, dairy, eggs) to supply leucine, carnitine precursors, and creatine naturally
- Include oily fish regularly for EPA (sardines, mackerel, salmon, tuna)
- Ensure adequate caloric density in small, frequent meals given common anorexia
- Aim for 1.0–1.5 g protein per kg body weight per day, per ESPEN guidelines, with evidence supporting the higher end during active cachexia
- **Work with a registered dietitian** to monitor intake and supplement gaps systematically
Interactions With Cancer Treatment
Some nutrients may improve chemotherapy response or reduce treatment-related toxicity. ESPEN guidelines support long-chain omega-3 supplementation to stabilize or improve appetite, food intake, lean body mass, and body weight in patients undergoing chemotherapy. Carnitine may similarly help reduce fatigue and toxicity in certain regimens.
By contrast, evidence for glutamine in preventing oral mucositis remains inconclusive, with current data insufficient to support routine use. Patients should discuss all nutritional interventions with their oncology team to confirm safety and avoid interference with their specific treatment protocol.
Frequently Asked Questions
Can nutrition alone reverse cancer cachexia?
Cancer cachexia cannot be fully reversed by caloric supplementation alone — metabolic disruptions driven by tumor-secreted factors and systemic inflammation continue regardless of caloric intake. Targeted nutritional interventions can slow progression and improve muscle function, but work best as part of a multimodal strategy that includes exercise and medical management.
What is the difference between cancer cachexia and regular weight loss?
In regular weight loss, the body reduces energy expenditure as a compensatory response. In cachexia, tumor-secreted factors and inflammation drive continuous catabolism — the body actively breaks down muscle protein regardless of how much the patient eats.
When should nutritional intervention for cancer cachexia begin?
Intervention is most effective in the pre-cachexia stage before significant muscle mass is lost. Regular evaluation of nutritional intake, weight change, and BMI should begin at cancer diagnosis and be repeated depending on clinical stability. Waiting until advanced weight loss has occurred dramatically reduces the impact of any intervention.
How much protein do cancer patients with cachexia need?
ESPEN guidelines suggest 1.0–1.5 g of protein per kg of body weight per day for cancer patients, with some evidence supporting the higher end during active cachexia. Prioritizing leucine-rich, high-quality protein sources (animal proteins, dairy, whey) is important alongside adequate total caloric intake to maximize muscle protein synthesis.
What supplements are most studied for cancer cachexia?
EPA (omega-3), HMB (β-hydroxy-β-methylbutyrate), L-carnitine, and leucine/BCAAs have the strongest clinical evidence for cachexia support. All four show better results when started early, used at established doses — EPA 2 g/day, HMB 3 g/day, carnitine 3–4 g/day — and combined rather than used alone.
Are there dietary strategies that specifically target cancer cell metabolism?
Yes — approaches like methionine restriction aim to exploit cancer cells' high dependence on methionine for growth, selectively stressing tumor cells while supporting normal tissue metabolism. This is distinct from general caloric restriction and represents an active area of research in integrative oncology, forming the foundation of protocols like NORI's cycled methionine-restriction program.
Moving Forward
Cancer cachexia is a metabolic syndrome — one that requires targeted intervention well beyond caloric increases. The evidence consistently points to early screening, multi-nutrient combinations, and exercise integration where tolerated. For patients seeking comprehensive support, programs combining dietary modification with evidence-based nutraceuticals and professional guidance produce the strongest outcomes for preserving muscle and maintaining function during cancer care.
For questions about nutritional support tailored to your specific cancer circumstances, contact NORI at 800-634-3804 or info@nutritionaloncology.net for a free initial consultation.


