
Low Methionine Diet & Cancer: Can It Help Treatment?
Most cancer patients focus solely on treatments like chemotherapy or surgery, unaware that a single dietary change — limiting one amino acid — may actively support those treatments. Research shows that the vast majority of cancer cells cannot survive without a constant external supply of methionine from food. This creates a metabolic vulnerability that researchers are targeting.
Unlike healthy cells, which can recycle methionine through normal metabolic pathways, cancer cells have lost this ability due to oncogenic mutations. What this means for patients is worth understanding: restricting dietary methionine may slow tumor growth and make conventional treatments work harder.
Here's what the science shows — and what it means for integrating this strategy into cancer care.
TLDR
- Cancer cells are uniquely dependent on dietary methionine, while most healthy cells can recycle it from homocysteine
- Methionine restriction has slowed tumor growth and enhanced chemotherapy and radiation effects in preclinical research
- Small human pilot studies confirm methionine-restricted diets are feasible and well-tolerated, though large clinical trials are still needed
- A primarily plant-based diet low in animal protein forms the practical foundation of methionine restriction
- Methionine restriction works best as a complementary strategy under guidance, not as a replacement for medical treatment
Why Cancer Cells Are "Addicted" to Methionine
Methionine is an essential amino acid your body cannot produce on its own; it must come from food. It plays critical roles in:
- Protein synthesis
- DNA methylation via S-adenosylmethionine (SAM)
- Glutathione production (central to cellular redox regulation)
- Cellular energy metabolism
The Hoffman Effect: A Cancer Cell Defect
In 1974, researchers discovered something remarkable: when methionine was replaced with its metabolic precursor homocysteine in growth media, malignant cells died while normal adult mammalian cells survived. This phenomenon, known as methionine dependence or the "Hoffman Effect," occurs because cancer cells have lost the ability to recycle methionine from homocysteine through the methionine cycle, despite retaining the enzymatic machinery to do so.
Research evaluating 23 diverse human tumor cell lines found that 11 were absolutely methionine-dependent (failed to grow entirely in homocysteine-substituted media) and 3 grew only slightly. This high frequency suggests methionine dependence is a common metabolic defect across cancers.
Why Cancer Cells Need So Much Methionine
Rapidly proliferating cancer cells need methionine in large quantities. They rely on SAM to fuel the constant DNA and histone methylation that sustains their abnormal gene expression patterns. Research shows cancer cells exhibit elevated rates of transmethylation (methyl group transfer) compared to normal fibroblasts — a demand healthy cells simply don't share.
A Vulnerability Across Many Cancer Types
Methionine dependence has been documented across:
- Prostate cancer
- Breast cancer (including triple-negative)
- Colorectal cancer
- Bladder cancer
- Glioma
- Melanoma
- Leukemia
- Sarcoma
- Kidney cancer
Even fresh patient tumor samples cultured outside the body demonstrate this vulnerability, making methionine restriction a potentially universal cancer target.
This selectivity — cancer cells dying while healthy cells continue to proliferate — is the biological basis for using dietary methionine restriction as a therapeutic strategy. The practical question is how deeply that restriction needs to go, and for how long, to meaningfully starve tumors without harming the patient.
How Methionine Restriction Attacks Tumors
Trapping Cancer Cells in the Cell Cycle
Methionine restriction causes cancer cells to become trapped in the late S/G2 phase of cell division, halting their proliferation while leaving normal cells largely unaffected. This arrest creates an opportunity: chemotherapy drugs that target specific cell cycle phases become far more effective when cancer cells are concentrated in the vulnerable phase.
In prostate cancer cells, methionine restriction caused arrest predominantly in the G2/M phase, accompanied by accumulation of cyclin-dependent kinase inhibitors p21 and p27, leading to apoptosis.
Depleting Cancer Cells' Pro-Oxidant Threshold
Cancer cells rely heavily on methionine to produce glutathione, their primary defense against oxidative damage. When methionine is restricted, glutathione is depleted, flooding cancer cells with reactive oxygen species (ROS) within 3-6 hours.
Cancer cells are known to have elevated oxidative stress — pro-oxidants are being investigated for how they may interact with these cellular conditions. Pro-oxidant therapy is being studied for its role in cellular stress pathways. Normal cells handle redox balance differently: methionine restriction activates NRF2, which restores redox balance through protective pathways — a response being researched in the context of cancer cells.
Targeting Cancer Stem Cells
That oxidative vulnerability extends even further in cancer stem cells. This small subpopulation drives treatment resistance and relapse, and is even more methionine-dependent than regular cancer cells. Their self-renewal depends on continuous SAM biosynthesis for histone methylation.
Research shows methionine restriction significantly reduces cancer stem cell populations and their tumor-forming capacity. In triple-negative breast cancer models, methionine restriction inhibited mammosphere formation and reduced the CD44hi/CD24low cancer stem cell population. Effects were partly rescued by SAM supplementation, confirming the mechanism.
Disrupting Malignant Gene Expression
Reduced SAM availability strips cancer cells of key histone methylation marks (H3K4me2, H3K4me3, H3K27me3) that maintain their malignant programming. This epigenetic destabilization is specific to cancer cells because their heightened transmethylation activity makes them far more sensitive to SAM depletion than normal cells.
Synergy With DNA-Damaging Treatments
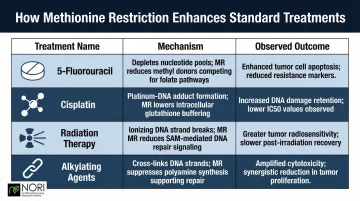
Methionine restriction augments chemotherapy and radiation through multiple mechanisms:
| Treatment | Mechanism | Observed Outcome |
|---|---|---|
| 5-Fluorouracil (5-FU) | Reduces 5,10-methylene-tetrahydrofolate by 75%; inhibits thymidylate synthase | Imbalanced nucleotide pool amplifies 5-FU cytotoxicity |
| Cisplatin | Increases intratumoral platinum accumulation | 32.1% T/C ratio in breast carcinoma mouse model |
| Radiation | Sensitizes RAS-driven tumor cells to focal radiation | Tumor tripling time extended 52% (17.48 → 26.57 days) with 20 Gy dose |
| Alkylating Agents | Down-regulates MGMT, a DNA-repair protein driving alkylating agent resistance | 13% decrease in MGMT activity in peripheral blood cells with cystemustine |
What the Research Actually Shows
The Landmark 2019 Nature Study
Research published in Nature in 2019 provided the strongest evidence to date for methionine restriction in cancer. Dietary methionine restriction (0.12% methionine w/w) synergized with 5-FU in colorectal cancer models and with radiation in a RAS-driven sarcoma model. The mechanism operated through tumor-cell-autonomous disruptions to one-carbon, redox, and nucleotide metabolism.
The same study included a human feasibility component: a controlled feeding study in 6 healthy middle-aged individuals used a low-methionine diet (~2.92 mg/kg/day, an 83% reduction from baseline) for 3 weeks. The diet reproducibly suppressed plasma methionine levels and altered circulating metabolism similarly to mouse models, demonstrating tolerability.
Human Pilot Studies Confirm Feasibility
A Phase I trial in 8 patients with metastatic solid tumors prescribed 2 mg methionine/kg/day. Patients remained on the diet for an average of 17.3 weeks. Plasma methionine fell from 21.6 ± 7.3 µM to 9 ± 4 µM within 2 weeks (a 58% decline). The primary side effect was weight loss of ~0.5 kg per week.
A feasibility study in 11 patients with metastatic colorectal cancer combined a 3-day methionine-free diet with the FOLFOX6 regimen. Plasma methionine was depleted by 58% on the first day of the diet. While feasible and well-tolerated, investigators noted the difficulty of administering the combination over multiple months.
Clinical Trials Landscape
These feasibility findings prompted several small clinical trials testing methionine restriction alongside standard therapies:
Recurrent glioblastoma + temozolomide: Phase 1 trial (18 patients) using a methionine-free diet (Hominex-2) for 7 days alternating with temozolomide. Completed in 2008.
Non-skin malignancies + radiation: Phase 1 pilot study (9 enrolled, 5 completed). Target plasma methionine was 13 µM; average nadir achieved was 16.8 µM. No grade 3+ adverse events occurred.
Metastatic triple-negative breast cancer + TRAIL agonist: Phase 2 trial (4 enrolled) evaluated intermittent methionine restriction with ONC201. Completed in 2021.
The Honest Bottom Line on Evidence
Here is where the evidence currently stands:
- Preclinical data is mechanistically compelling across multiple cancer types and treatment combinations
- Human feasibility is established, with consistent plasma methionine reductions across trials
- The critical gap is the absence of large randomized controlled trials with clinical endpoints
Researchers note the main barriers have been dietary adherence challenges, palatability issues with methionine-free medical foods, and recruitment difficulties. One trial was closed early because "the diet was not desirable for most patients who were offered the trial and refused, and was not acceptable for most of the patients enrolled."
Ongoing work is focused on improving dietary delivery — better-tasting medical foods and cycling protocols — to make methionine restriction practical enough to test at scale.
What a Low Methionine Diet Looks Like in Practice
Foods to Minimize
Animal proteins are the primary methionine contributors:
- Red meat: Beef contains ~662 mg methionine per 100g
- Poultry: Chicken breast contains ~585 mg per 100g
- Fish and shellfish: Salmon provides ~785 mg per 100g; shellfish range similarly high
- Eggs: Whole eggs contain ~392 mg per 100g
- Dairy products: Parmesan and hard cheeses are among the highest dairy sources
Patients following a methionine-restricted protocol typically replace the majority of protein intake with low-methionine plant foods, with only a small fraction coming from animal sources, if any.
Foods to Emphasize
Low-methionine, nutrient-dense plant foods include:
- Fruits: Very low methionine content (~2 mg per medium apple)
- Non-starchy vegetables: Most contain under 20 mg per 100g — eat freely
- Legumes: Firm tofu contains ~211 mg per 100g — significantly lower than animal proteins
- Whole grains: Oats (~182 mg per 100g) are a practical staple
- Select soy foods: Edamame and tempeh offer protein without the methionine load of meat

A well-designed low-methionine diet must still meet total protein and calorie needs. ESPEN guidelines recommend 1.0 to 1.5 g/kg/day for cancer patients to prevent muscle wasting and cachexia.
Inadequate protein is a genuine risk during cancer treatment. Muscle preservation matters — and getting that balance right without professional input is harder than it looks.
Structured Guidance Makes a Difference
Self-directed dietary restriction is difficult to implement correctly. An unguided approach risks nutritional deficiencies while failing to achieve meaningful methionine reduction — the two problems most patients encounter when trying this alone.
NORI's home-based nutritional support program uses a personalized cycled methionine restriction approach, combining strategic dietary phases with nutraceutical support and continuous guidance to help patients stay on track without compromising their health. For a free consultation, call 800-634-3804.
Safety Considerations, Limitations, and Who May Benefit Most
Essential Safety Caveats
Methionine is necessary for immune function, muscle protein synthesis, and neurological health. T-cells require methionine to mount immune responses. Severe or prolonged restriction without proper nutritional planning can cause muscle wasting, immune suppression, and other deficiencies.
Patients should always pursue methionine restriction under the supervision of a qualified healthcare professional and never sacrifice adequate total nutrition.
Critical Limitation Regarding Immunotherapy
Emerging evidence suggests methionine restriction may impair T-cell function and immunotherapy response. Two specific mechanisms have been documented:
- Methionine restriction reduces histone H3K4me3 at promoter regions of key genes required for T-cell proliferation
- Sustained methionine restriction induces upregulation of T-cell exhaustion markers
The picture is more nuanced with intermittent restriction. Intermittent dietary methionine deprivation has been investigated for its effects on cellular health pathways and potential synergy with PD-1 checkpoint blockade in preclinical models. Yet in immunocompetent patients, continuous restriction may become pro-cancer by impairing T-cell activation.
Patients receiving checkpoint inhibitors or other immunotherapy should discuss the potential conflict with their oncologist before attempting methionine restriction.
Who May Benefit Most
Current research points to four groups where methionine restriction shows the clearest rationale:
- DNA-damaging therapy patients — Research supports methionine restriction as a synergistic add-on for those receiving 5-FU, cisplatin, doxorubicin, or radiation, given the documented mechanism overlap.
- Advanced or late-stage cancers — Patients where even modest improvements in treatment sensitivity matter may have the most to gain.
- Tumors with MTAP loss — Tumors lacking Methylthioadenosine Phosphorylase (occurring in ~15% of cancers) cannot recycle methionine from the polyamine pathway, making them exceptionally sensitive to exogenous restriction.
- Patients who have exhausted conventional options — When standard treatments have failed, the risk-benefit balance may favor a well-supervised nutritional intervention that targets a documented cancer metabolic vulnerability.

Frequently Asked Questions
Can stage 4 cancer achieve wellness improvements?
Yes, stage 4 cancer patients have reported client-reported wellness improvements in some cases — outcomes depend on cancer type, individual biology, and treatment approach. Methionine restriction is one integrative strategy being explored to support conventional treatment outcomes, including in advanced disease.
What foods are highest in methionine that cancer patients should limit?
Red meat, poultry, fish, eggs, and dairy are the primary high-methionine foods. A low-methionine dietary approach shifts the protein base primarily toward plant foods such as fruits, vegetables, legumes, and grains.
Is a low methionine diet safe to follow during chemotherapy?
A properly designed methionine-restricted diet that still meets total calorie and protein needs has been shown to be well-tolerated in small human studies alongside chemotherapy. However, supervision by a healthcare professional is essential to avoid nutritional deficiencies or muscle loss.
How is methionine restriction different from a vegan or plant-based diet?
A vegan diet naturally contains less methionine than animal-protein diets, but a therapeutic methionine restriction protocol is more deliberate. It is specifically formulated — using precise dietary guidelines or supplemental medical foods — to reduce plasma methionine to levels that measurably affect cancer cell metabolism.
Does methionine restriction work for all types of cancer?
Methionine dependence has been documented across a wide range of cancer types in laboratory and animal studies, including breast, prostate, colorectal, glioma, and melanoma. However, the degree of dependence may vary and not all cancers are equally responsive, which is why research continues to investigate which tumor types and treatment combinations benefit most.
Can a low methionine diet replace chemotherapy or radiation?
Current evidence does not support methionine restriction as a standalone cancer treatment — its value lies in its potential to enhance the effectiveness of conventional DNA-damaging therapies. It is best understood as a complementary strategy within a broader treatment plan, not as a replacement.


