
While "eat better" is commonly advised, the biological mechanisms behind these approaches remain poorly understood. Calorie restriction (CR) reduces circulating glucose and growth hormones that fuel cancer cells. Anti-inflammatory diets lower systemic inflammation, one of the key environmental conditions allowing cancer to initiate and progress. This article explains the science behind these strategies, their measurable benefits, and how to apply them—not just in theory, but in practice.
TL;DR
- Calorie restriction deprives cancer cells of glucose and growth hormones, creating metabolic stress that normal cells survive but cancer cells cannot
- Anti-inflammatory diets reduce chronic inflammation—a key driver of DNA damage and tumor-permissive microenvironments
- Combining both approaches targets cancer metabolism and inflammatory pathways together, compounding protective effects
- Recent research shows dietary restriction enhances T cell immune function by fueling T cells with ketones, boosting health-supporting immunity
What Are Calorie Restriction and Anti-Inflammatory Diets?
Calorie restriction in cancer research refers to a sustained 20–40% reduction in daily caloric intake that maintains adequate nutrition while depriving cancer-promoting metabolic pathways of excess energy. This is distinct from malnutrition or crash dieting, which have harmful effects on immunity. Studies like CALERIE demonstrate that a 25% calorie reduction target improves metabolic health without causing nutrient deficiencies.
Anti-inflammatory dietary patterns—such as Mediterranean, DASH, and Nordic diets—emphasize whole foods, colorful plant matter, healthy fats, and lean proteins while eliminating ultra-processed foods, added sugars, and red and processed meats. These frameworks work as modifiable metabolic environments, shifting the body's internal conditions from cancer-permissive to cancer-resistant rather than functioning as short-term fixes.
Each pattern takes a distinct approach to achieving this shift:
Key characteristics of anti-inflammatory dietary patterns:
- Mediterranean Diet: Prioritizes olive oil, fruits, nuts, vegetables, legumes, and whole grains; includes moderate fish and poultry; limits red and processed meats
- DASH Diet: Centers on fruits, vegetables, nuts, seeds, legumes, and fiber-rich cereals; restricts sodium, sugar-sweetened beverages, and red or processed meat
- Nordic Diet: Focuses on fruit, vegetables, whole grains, and fish; substitutes rapeseed (canola) oil for olive oil

Together, these patterns form a practical foundation for understanding how dietary choices interact with the metabolic conditions that either support or suppress cancer development.
How These Dietary Strategies Fight Cancer at the Cellular Level
Exploiting the Warburg Effect
Cancer cells rely almost exclusively on glucose for energy through a process called aerobic glycolysis—the Warburg Effect. This metabolic shift makes them uniquely vulnerable to calorie restriction and sugar reduction. When glucose is limited, normal cells adapt by switching to ketones for fuel. Cancer cells, however, cannot efficiently use ketones due to low expression of ketolytic enzymes like BDH1 and OXCT1, creating a metabolic disadvantage for tumors.
Activating Autophagy via AMPK and mTOR Pathways
Calorie restriction activates autophagy, a cellular housekeeping process that degrades damaged organelles and clears pre-cancerous cell components. Under nutrient deprivation, AMPK activates and directly phosphorylates ULK1, the kinase that initiates autophagy. Simultaneously, CR reduces insulin and IGF-1 signaling, which suppresses the PI3K/AKT/mTORC1 pathway. Because mTORC1 normally inhibits autophagy, its suppression through CR further enhances this protective process.
CR-induced autophagy serves critical quality-control functions:
- Mitophagy: Selectively clears damaged mitochondria that generate carcinogenic reactive oxygen species
- Protein aggregate removal: Eliminates misfolded proteins that can contribute to oncogenic transformation
- DNA repair support: Frees cellular resources to improve DNA repair efficiency and reduce free radical-induced damage
Reducing Chronic Inflammation
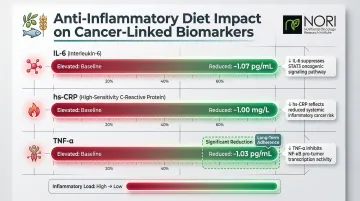
When pro-inflammatory cytokines, particularly IL-6 and TNF-α, remain chronically elevated, they damage healthy cells, suppress immune surveillance, and create a tumor-permissive microenvironment. These cytokines activate the NF-κB transcription factor, which upregulates anti-apoptotic genes and drives cancer cell proliferation. Anti-inflammatory diets directly reduce these cytokine levels.
Research demonstrates that Mediterranean dietary patterns significantly lower inflammatory biomarkers:
- IL-6 reduced by 1.07 pg/mL on average
- hs-CRP reduced by 1.00 mg/L on average
- TNF-α significantly reduced in long-term studies
These reductions directly counteract the inflammatory pathways (IL-6/STAT3 and TNF-α/NF-κB) that drive cancer progression.
Reshaping the Tumor Microenvironment
Preclinical studies show that a 40% calorie restriction regimen significantly decreased tumor vascularity and reduced VEGF expression — the protein that signals new blood vessel formation — effectively starving tumors of their blood supply.
Recent research reveals how CR enhances immune function within the tumor microenvironment. A 2025 study in Nature Metabolism from the Van Andel Institute showed that dietary restriction promotes ketone production, which acts as fuel specifically for T cells, enhancing their health-supporting effectiveness and preventing cellular exhaustion. The research demonstrates that CD8+ T cells oxidize ketones to boost mitochondrial function, fuel the TCA cycle, and promote expansion of highly functional effector T cells.
These findings are promising, though tumor heterogeneity complicates the picture. While most tumors are vulnerable to ketosis due to low ketolytic enzyme expression, some cancers can upregulate these enzymes and exploit ketones for growth — underscoring why individualized dietary protocols matter.
Key Benefits of Combining Both Dietary Approaches
While calorie restriction and anti-inflammatory eating each offer independent health-supporting benefits, their combined effect is synergistic—targeting cancer metabolism, its inflammatory microenvironment, and immune response simultaneously.
Disrupting Cancer Cell Metabolism and Growth Signaling
Reducing caloric intake—particularly sugars and refined carbohydrates—lowers circulating insulin and IGF-1 (insulin-like growth factor 1), key anabolic hormones that stimulate cancer cell proliferation. When these signaling pathways (PI3K/AKT/mTOR) are suppressed, cancer cell growth slows.
This creates what researchers call "differential stress resistance": normal cells adapt to reduced nutrient availability by entering maintenance mode, while cancer cells—unable to slow down—may become more responsive to treatment. Research confirms that fasting or fasting-mimicking diets combined with chemotherapy reduce treatment-related toxicity in normal cells, with research into cellular stress pathway effects being explored alongside conventional care.
This metabolic disruption translates to reduced tumor growth rates, enhanced chemotherapy sensitivity, and better protection of healthy cells during treatment. The effect is strongest for metabolically driven cancers (breast, colorectal, prostate) and for patients who are pre-diabetic or insulin-resistant, where excess calories actively accelerate cancer-permissive signaling.
Key biological markers affected: circulating insulin, IGF-1, glucose levels, mTOR activation, tumor growth rate
Reducing Chronic Inflammation That Enables Cancer to Thrive
Chronic inflammation is not just a symptom but an active driver of carcinogenesis. It creates DNA damage, promotes abnormal cell survival, affects cellular stress pathways, and creates a microenvironment that aids tumor invasion and metastasis. Anti-inflammatory diets directly interrupt this cycle.
Eliminating ultra-processed foods and added sugars reduces NF-κB pathway activation—a master inflammatory signaling hub. Increasing omega-3 fatty acids, polyphenols, and colorful plant foods elevates anti-inflammatory compounds including resveratrol, curcumin, and quercetin, which are also studied as caloric restriction mimetics (CRMs—compounds that replicate the metabolic effects of calorie reduction).
Meta-analyses demonstrate that high adherence to Mediterranean dietary patterns significantly reduces cancer incidence:
- Colorectal cancer: 16% risk reduction (HR: 0.84)
- Breast cancer: 13% risk reduction (HR: 0.87)
- Overall cancer mortality: 13% risk reduction (RR: 0.87)
This benefit is especially relevant for individuals with pre-existing inflammatory conditions, obesity, or high-sugar dietary patterns—and for cancer survivors working to reduce recurrence risk. Markers that shift with these dietary changes include CRP, IL-6, TNF-α, NF-κB activity, and DNA damage indicators.

Strengthening Immune Surveillance Against Cancer Cells
One of the body's primary defenses against cancer is immune surveillance—the ability of T cells and natural killer (NK) cells to identify and destroy abnormal cells before they form tumors. Both chronic inflammation and metabolic dysfunction impair this process.
Calorie restriction strengthens this defense through several mechanisms: it raises CD8+ cytotoxic T cell counts, reduces immunosuppressive regulatory T cells (Tregs) in the tumor microenvironment, and supplies T cells with ketones as an alternative metabolic fuel. That fuel supply helps T cells stay active rather than exhausting prematurely. Anti-inflammatory foods rich in resveratrol and curcumin further support NK cell activity.
Preclinical research suggests that combining calorie restriction with anti-inflammatory eating may amplify immune checkpoint inhibitors by keeping T cells metabolically primed. This makes the combined approach particularly worth considering for:
- Patients currently undergoing or considering immunotherapy
- Individuals with suppressed immunity or a family history of cancer
- Cancer survivors focused on long-term immune health
Biological markers that reflect these shifts include CD8+ T cell counts, NK cell activity, Treg suppression, and tumor immune infiltration.
What Happens When Chronic Inflammation and Excess Caloric Intake Go Unchecked
Without dietary intervention, chronic inflammation and elevated insulin/IGF-1 from excess caloric intake create a progressively more cancer-permissive environment. DNA damage accumulates, immune surveillance declines, and tumor microenvironments become increasingly favorable to cancer cell survival.
The statistical burden is substantial. According to the IARC, excess body weight is attributable to approximately 500,000 new cancer cases globally in 2022. In the United States, 2019 data shows that excess body weight accounted for 10.6% of cancer cases in women and 4.8% in men, while suboptimal diet contributed to 4.9% of cases in men and 3.4% in women.
Those numbers reflect a clear biological pattern. Obesity is strongly associated with increased risk for at least 13 cancer types — including endometrial, esophageal adenocarcinoma, colorectal, and postmenopausal breast cancers — driven by three overlapping mechanisms:
- Chronic low-grade inflammation that promotes DNA damage
- Elevated insulin and IGF-1 signaling that accelerates cell proliferation
- Altered sex hormone levels that fuel hormone-sensitive cancers
The missed opportunity in conventional care:
Most standard oncology protocols do not address diet as part of treatment. Surveys indicate that up to 35% of outpatient cancer centers do not use validated malnutrition screening tools, and the registered dietitian-to-patient ratio is severely inadequate (averaging 1:2,308).
Patients relying solely on drugs, radiation, or chemotherapy miss the additional protection that targeted nutritional intervention can provide. Integrative programs that combine dietary restriction with nutraceutical support can help close that gap — addressing the metabolic and inflammatory conditions that conventional treatment leaves largely untouched.
How to Get the Most Value from These Dietary Strategies
Both approaches work best when applied consistently and in combination. Calorie restriction without anti-inflammatory food choices can still allow inflammatory damage. Anti-inflammatory eating without managing overall caloric load may leave cancer-promoting insulin and IGF-1 elevated. The full protective effect requires both elements working together.
Advanced Application: Methionine Restriction
Beyond general calorie restriction, restricting the amino acid methionine offers a more targeted nutritional approach. Methionine restriction is being studied in nutritional oncology research for cancer cells, which are known to have a higher dependency on methionine than normal cells—a phenomenon known as the "Hoffman effect." Research suggests cancer cells may experience distinct metabolic responses when methionine is restricted, while normal cells can tolerate low methionine by remethylating homocysteine.
This principle forms the foundation of NORI's protocol, which cycles methionine restriction alongside proprietary nutraceutical combinations to support metabolic wellness. Unlike broad dietary guidelines, this approach targets a metabolic pattern specific to cancer cells that is being investigated in nutritional oncology research.
High-methionine foods to limit include:
- Animal proteins: Ground turkey (931 mg per 100g), beef (905 mg per 100g), tuna (885 mg per 100g)
- Plant proteins and dairy: Brazil nuts (1,124 mg per 100g), firm tofu (211 mg per 100g), parmesan cheese

Personalized Guidance and Professional Support
Dietary needs vary significantly based on cancer type, treatment status, and individual metabolic response. A practitioner reviews outcomes regularly and adjusts the approach as circumstances change—something a static diet plan cannot do.
NORI's home-based nutritional support program provides this kind of ongoing, individualized care. Key features include:
- Unlimited consultation support throughout the program
- Customized diet plans and nutraceutical protocols tailored to each patient
- Ongoing adjustments for patients in active conventional treatment
This oversight matters especially for patients at risk of malnutrition or cachexia, where implementing dietary restriction without guidance can cause harm rather than benefit.
Conclusion
Calorie restriction and anti-inflammatory diets address cancer prevention from two of the most scientifically validated pathways: disrupting cancer metabolism and dismantling the inflammatory conditions cancer depends on. Their value lies not in any single intervention but in the sustained, combined approach.
These strategies compound over time. The longer they are applied consistently, the more protective the effects become—through improved immune surveillance, reduced tumor microenvironment favorability, and lower baseline inflammation. Biomarkers like insulin, CRP, and inflammatory cytokines can shift within weeks, but the protective effects on cancer risk accumulate over months and years of consistent practice.
The three mechanisms driving this protection are worth keeping in mind:
- Metabolic disruption: Calorie restriction starves cancer cells of the glucose and growth signals they depend on
- Inflammation reduction: Anti-inflammatory dietary patterns remove the chronic tissue environment that enables tumor initiation and progression
- Immune restoration: Both approaches improve the body's ability to detect and clear abnormal cells before they establish
Diet isn't a passive background factor in cancer risk. Applied deliberately and consistently, it becomes one of the most accessible tools for long-term prevention.
Frequently Asked Questions
Can calorie restriction support cancer wellness from developing in the first place?
Preclinical and emerging clinical evidence supports CR's role in reducing cancer initiation by lowering insulin/IGF-1 levels, reducing oxidative stress, and enhancing immune surveillance. However, it reduces risk rather than guaranteeing prevention.
What foods should I eliminate first to reduce cancer-promoting inflammation?
Added sugars, ultra-processed foods, red and processed meats, and refined carbohydrates are the most impactful categories to reduce first. They are primary drivers of NF-κB-mediated chronic inflammation and elevated insulin signaling.
Is calorie restriction safe for cancer patients who are already undergoing chemotherapy?
CR may offer benefits like differential stress resistance and reduced side effects, but it also carries real risks of malnutrition, cachexia, and muscle loss in active cancer patients. Any dietary restriction during treatment requires professional guidance matched to the individual's nutritional status.
How is methionine restriction different from general calorie restriction?
General CR reduces overall caloric intake, while methionine restriction targets a specific amino acid that cancer cells rely on far more heavily than healthy cells. This selectivity makes it a more precise nutritional strategy, and it forms the core mechanism of the NORI Protocol.
Can an anti-inflammatory diet help even if I already have cancer?
Anti-inflammatory dietary patterns may improve treatment tolerance, reduce inflammatory cytokines that support tumor growth, and improve quality of life for people already diagnosed with cancer. It should be used as a complement to, not replacement for, a structured treatment plan.
How long does it take for dietary changes to meaningfully affect cancer risk?
While some biomarkers (insulin, CRP, inflammatory cytokines) can shift within weeks of dietary change, the protective effects on cancer risk accumulate over months and years of consistent practice. Sustained patterns over 6–12 months carry far more weight than any single period of strict compliance.


