
Introduction
More than 35% of cancer patients actively explore dietary interventions to complement or, in some cases, replace conventional treatment. The central question driving this surge: can changing what you eat actually starve cancer cells while protecting healthy ones?
The premise stems from a century-old discovery. In 1924, Otto Warburg observed that tumors consume glucose at an abnormally high rate even when oxygen is plentiful, a pattern now recognized as one of cancer's defining hallmarks.
This metabolic vulnerability has driven a generation of dietary strategies designed to exploit what cancer cells depend on most: specific nutrients like glucose and certain amino acids.
Yet the science is far from settled. Emerging evidence reveals both promise and peril—including recent high-profile studies showing that glucose restriction can paradoxically promote metastasis under certain conditions. This article examines the science behind metabolic therapy diets, the main approaches (ketogenic, fasting, and methionine restriction), the clinical evidence, the serious risks involved, and how to make informed decisions.
TLDR
- Cancer cells depend heavily on glucose and specific amino acids (especially methionine), creating dietary vulnerabilities that targeted nutritional strategies can exploit
- Ketogenic diets, intermittent fasting/caloric restriction, and methionine restriction are the primary metabolic strategies studied for cancer
- Preclinical evidence is substantial, but human trials remain limited by small sample sizes and few large randomized controlled studies
- Critical 2025 research warns glucose restriction may suppress primary tumors but can promote lung metastasis in certain conditions
- No single metabolic approach works in isolation—personalized protocols under expert supervision reduce risk and improve outcomes
What Is Metabolic Therapy for Cancer?
Metabolic therapy in oncology represents a class of interventions targeting the altered energy metabolism of cancer cells to impair their growth, survival, or resistance to treatment. This differs fundamentally from simply "eating healthy." Instead of general wellness nutrition, metabolic therapy deliberately manipulates specific fuel substrates to create an environment hostile to cancer cells while leaving healthy cells relatively unharmed.
The Warburg Effect: Why Cancer Cells Are Metabolically Different
In 1924, Otto Warburg first described what is now known as the Warburg Effect—cancer cells' tendency to rely on aerobic glycolysis, breaking down glucose into lactate even when oxygen is present. Normal cells use the far more efficient oxidative phosphorylation process when oxygen is available, but cancer cells bizarrely prefer the wasteful glycolysis pathway.
This metabolic rewiring was formally recognized as a hallmark of cancer in 2011 under the category "deregulating cellular energetics." Pan-cancer studies confirm this phenomenon is universal—all tumors show increased glycolysis levels regardless of their tissue of origin.
This glucose dependence creates a selective vulnerability. Healthy cells maintain metabolic flexibility—they can switch fuel sources when glucose is scarce. Cancer cells, locked into glycolysis, largely cannot. Certain tumors compound this rigidity by also requiring unusually high amounts of the amino acid methionine for DNA methylation and rapid cell division, creating additional targets beyond carbohydrates alone.
How Metabolic Therapy Diets Exploit This Vulnerability
Restrict the specific nutrients cancer cells depend on—carbohydrates, glucose, or certain amino acids—and you impose metabolic stress that cancer cells, locked into rigid fuel preferences, struggle to survive. Healthy cells adapt by shifting to alternative fuels like ketone bodies or recycling amino acids more efficiently.
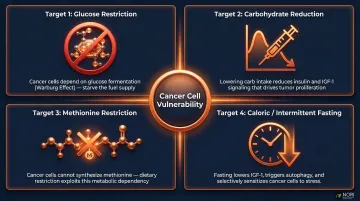
The main dietary targets metabolic therapy protocols address include:
- Glucose restriction — limits the primary fuel source for aerobic glycolysis
- Carbohydrate reduction — lowers circulating blood glucose and insulin, which drives tumor growth signaling
- Methionine restriction — starves tumors of the amino acid required for DNA methylation reactions and polyamine synthesis (compounds that support rapid cell division)
- Caloric or intermittent fasting protocols — compounds metabolic stress by reducing overall substrate availability
The Main Types of Metabolic Therapy Diets
Several distinct dietary approaches have been studied, each targeting different cancer metabolic vulnerabilities and suiting different patient profiles.
Ketogenic Diet
The ketogenic diet for cancer typically consists of:
- 70–90% fat (often using medium-chain triglycerides for better tolerability)
- Very low carbohydrate (2–6% of calories)
- Moderate protein (8–25% of calories)
The goal is inducing ketosis, shifting the body's primary fuel from glucose to ketone bodies—molecules many cancer cells cannot efficiently utilize due to mitochondrial dysfunction or low expression of ketolytic enzymes (BDH1 and OXCT1, the proteins responsible for breaking down ketones).
Proposed mechanisms:
- Starves glycolysis-dependent tumors by reducing blood glucose and insulin levels
- Fuels healthy cells with ketones while tumors with dysfunctional mitochondria cannot access them
- Clinical trials show fasting blood sugar drops from 100.4 to 84.5 mg/dL on ketogenic diets, with corresponding insulin reductions
Fasting and Caloric Restriction
Intermittent fasting and short-term caloric restriction create systemic metabolic stress by:
- Lowering insulin and IGF-1 levels (hormones associated with cancer progression)
- Reducing blood glucose availability
- Triggering autophagy (cellular "cleanup" processes)
The "differential stress sensitization" concept, pioneered by researchers like Valter Longo, proposes that fasting forces normal cells into a protective maintenance mode while highly proliferative cancer cells fail to adapt, becoming more vulnerable to chemotherapy.
The randomized DIRECT trial demonstrated that breast cancer patients receiving fasting-mimicking diets alongside chemotherapy had 4.1 times higher likelihood of achieving 90–100% pathological tumor cell loss compared to controls, with reduced DNA damage in healthy immune cells.
Methionine Restriction
Methionine restriction is one of the more targeted metabolic approaches — and one of the least discussed. Cancer cells require methionine, an essential amino acid, in disproportionately high amounts for:
- DNA synthesis and methylation reactions
- Polyamine production for rapid cell division
- Maintenance of cellular redox balance (glutathione synthesis)
This dependency is known as the "Hoffman Effect." Restricting dietary methionine to 10 mg/kg of body weight per day induces cell cycle arrest and apoptosis in cancer cells, while healthy cells compensate by recycling methionine more efficiently.
Implementation in practice:
- Plant-based, low-methionine diet reducing animal proteins, eggs, and methionine-rich nuts — reducing animal protein without causing malnutrition can support metabolic wellness, reduce IGF-1 levels, and is being explored alongside conventional care in ongoing research
- Cycled protocols (e.g., 7 days on restriction, 7 days off) to prevent adaptation
- Often combined with pro-oxidant nutraceuticals that may support the body's natural cellular processes
One real-world application of this approach is the NORI Protocol, which combines cycled methionine restriction with proprietary nutraceutical formulations (including sodium selenite, vitamin B6, and plant extracts) developed to address metabolic vulnerabilities common across cancer types.
What Does the Evidence Say?
The evidence landscape spans preclinical animal studies, small human trials, case reports, and emerging large-scale analyses—showing genuine promise alongside important limitations.
Preclinical Evidence: What Animal and Cell Studies Show
A 2021 systematic review and meta-analysis evaluated 38 animal studies, finding that ketogenic diets significantly prolonged survival time and reduced tumor weight/volume in the majority of models. A 2024 Bayesian analysis of 1,755 individual mouse survival times confirmed that ketogenic diet monotherapy has been associated with positive wellness outcomes in observational research.
However, results varied dramatically by cancer type and genetic background. Some tumor types showed no effect or even tumor-promoting effects—particularly BRAF V600E-positive melanomas, where ketone bodies directly fuel mutated MEK1 pathways, accelerating tumor growth.
Methionine restriction shows equally compelling preclinical data. Studies demonstrate that methionine-depleted diets inhibit tumor growth across colorectal, breast, and prostate cancer models, suppress proliferation markers, and enhance chemotherapy efficacy in patient-derived xenografts (human tumor cells implanted in mice).
Clinical Evidence: Human Trials and Case Reports
Human evidence remains limited but promising. Most data comes from pilot studies and small feasibility trials, predominantly in:
- Glioblastoma and brain cancers
- Ovarian and endometrial cancer
- Breast cancer
- Non-small cell lung cancer
Consistent findings across trials include:
- Successful induction of ketosis with modest blood glucose reduction
- Good overall tolerability with dropout rates of 20–50% due to dietary restrictions
- Preservation of lean mass in many patients
- Improved quality of life in some cases
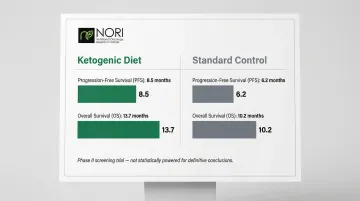
Notable randomized controlled trial: A phase II screening trial in metastatic pancreatic cancer patients combined ketogenic diet with gemcitabine, nab-paclitaxel, and cisplatin. Median progression-free survival was 8.5 months (keto) vs. 6.2 months (control), with overall survival of 13.7 months vs. 10.2 months—though not statistically powered for definitive conclusions.
Documented case reports showing tumor regression:
Zuccoli et al., 2010 documented a 65-year-old glioblastoma patient who showed no discernible tumor tissue on PET or MRI after 2.5 months of restricted ketogenic diet combined with standard therapy.
Nebeling et al., 1995 reported two pediatric astrocytoma patients on MCT-based ketogenic diets showing an average 21.8% decrease in glucose uptake at tumor sites via PET scan.
NORI methionine restriction case evidence:
Stage IV Papillary Thyroid Cancer: A published NORI case report documents a patient who reported thyroglobulin tumor markers dropping from 6.0 to 0.25 within two months of methionine restriction with sodium selenite. The patient reported significant improvement maintained over seven years.
Stage IV ER+ Breast Cancer: A patient reported CA15-3 tumor markers falling from nearly 1,000 to 250 within three months on the NORI protocol. Bone pain subsided completely using cycled methionine restriction combined with sodium selenite, vitamin K3, and genipin.
These cases reflect early-stage observational data—meaningful signals, but not yet the controlled trials needed to establish efficacy at a population level.
The State of the Evidence: Key Caveats
Despite promising signals, critical limitations remain:
- Small sample sizes (most trials include 20–80 patients)
- Heterogeneous study designs with no standardized dietary protocols
- High dropout rates due to dietary restrictions (20–50% in many trials)
- Lack of large randomized controlled trials powered for survival endpoints
- Short follow-up periods limiting long-term efficacy assessment
Larger, well-designed trials are underway—and the field is moving. Until those results emerge, metabolic dietary approaches are best understood as promising adjuncts rather than replacements for established treatment.
Important Risks and Limitations to Know
The Metastasis Paradox: A Critical Warning
Groundbreaking 2025 research published in Cell by Wu et al. uncovered a dangerous paradox: while glucose restriction suppresses primary tumor growth, it can simultaneously promote lung metastasis through a complex mechanism.
Glucose deprivation induces ER stress, triggering cancer cells to package TRAIL molecules into exosomes. These exosomal TRAIL molecules exhaust Natural Killer cells in the lungs, creating a "pre-metastatic niche" that enables cancer spread. A parallel 2024 study found ketogenic diets promote breast cancer metastasis by upregulating BACH1-mediated pro-metastatic gene transcription.
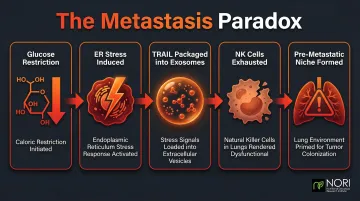
Both findings point to the same conclusion: tumor response to metabolic restriction varies by cancer type, stage, and individual biology — making expert oversight essential before starting any protocol.
Practical Risks
Micronutrient deficiencies: Long-term ketogenic diets frequently cause deficiencies in selenium, magnesium, phosphorus, iron, B vitamins, vitamin D, and calcium. Pediatric epilepsy studies document progressive bone mineral loss despite supplementation.
Cachexia and weight loss: Patients with existing malnutrition or low body weight face serious risks. Preclinical evidence links ketogenic diets to accelerated cachexia via corticosterone deficiency and NADPH depletion.
Fasting contraindications: European oncology nutrition guidelines strictly contraindicate fasting in underweight patients, those actively losing weight, or patients with diabetes or heart disease.
Methionine restriction challenges: A Phase I trial combining methionine restriction with radiation closed early — patients could not maintain the diet's demands, and accrual stalled.
The Need for Qualified Supervision
No metabolic therapy diet should begin without consulting an oncologist or qualified integrative nutritionist. These approaches function best as adjunctive tools within a comprehensive cancer management strategy—not as replacements for evidence-based standard care.
Is Metabolic Diet Therapy Right for You?
The Personalization Imperative
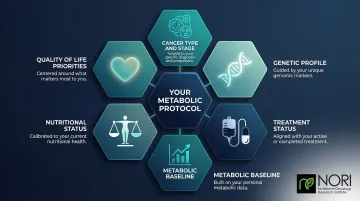
Research confirms that the right metabolic diet depends on individual factors — not universal protocols:
- Cancer type and stage
- Genetic profile (e.g., BRAF V600E mutation makes keto counterproductive in melanoma)
- Treatment status (active chemotherapy vs. surveillance)
- Metabolic baseline (how heavily your tumor relies on glucose vs. amino acids)
- Nutritional status (existing cachexia, body mass, micronutrient levels)
- Quality of life priorities
The field is moving toward patient-specific metabolic interventions guided by tumor metabolic phenotyping — meaning your oncology team or nutritional specialist should tailor any dietary protocol to your tumor's biology, not a one-size-fits-all template.
What a Personalized Evaluation Should Include
A thorough evaluation covers three areas:
Tumor metabolic signature:
- FDG-PET imaging to assess glucose dependency
- Ketolytic enzyme expression (OXCT1, BDH1) to predict ketone utilization
- Genetic profiling for contraindications (BRAF V600E status)
Current health status:
- Nutritional evaluation (body composition, micronutrient levels)
- Co-existing conditions (diabetes, heart disease, malnutrition)
- Treatment timeline (chemotherapy cycles, radiation schedule)
Quality of life priorities:
- Dietary restriction tolerance
- Symptom management goals
- Long-term sustainability considerations
Accessible Personalized Support
Working through these factors on your own can be difficult, particularly when tumor metabolic data isn't routinely ordered in standard oncology care. NORI (Nutritional Oncology Research Institute) offers home-based nutritional support programs that help patients implement protocols like methionine restriction alongside complementary nutraceuticals — no clinic visits required. Free initial consultations are available, with support accessible to patients worldwide.
Frequently Asked Questions
What is the diet for metabolic therapy?
Metabolic therapy diets include any dietary approach exploiting cancer cells' altered metabolism—most commonly the ketogenic diet (high-fat, very low-carb), intermittent fasting/caloric restriction, and methionine restriction (a plant-based low-amino acid approach). These are often combined with targeted nutraceuticals to amplify health-supporting effects.
What is the Warburg effect and why does it matter for cancer diet therapy?
The Warburg effect describes cancer cells' dependence on glycolysis for energy even when oxygen is present, making them disproportionately reliant on glucose. Metabolic diets exploit this dependency by restricting glucose and carbohydrates, creating an energy crisis cancer cells struggle to survive.
Can a metabolic therapy diet replace chemotherapy or conventional treatment?
No. Metabolic therapy diets are not proven replacements for standard cancer care. They function best as adjuncts that may enhance cancer care efficacy, reduce side effects, or support long-term management—always coordinated with your oncology team.
Is the ketogenic diet safe for all cancer patients?
No. Ketogenic diets may be contraindicated in certain cancer types (especially BRAF V600E-positive melanomas), can worsen cachexia in malnourished patients, and emerging evidence suggests they may promote metastasis under specific conditions. Individualized medical evaluation is essential before starting any ketogenic protocol.
What is methionine restriction, and how does it target cancer?
Methionine restriction limits an essential amino acid (found primarily in animal proteins) that many cancer cells require in excess for DNA replication and cell division. Healthy cells adapt by recycling methionine more efficiently, creating a selective metabolic disadvantage for cancer cells.
How long does it take to see results from a metabolic therapy diet for cancer?
Timelines vary widely by cancer type, stage, dietary approach, and concurrent therapies. Published case reports show responses ranging from 2.5 months (glioblastoma on ketogenic diet) to 10 months (chronic lymphocytic leukemia on methionine restriction).


