
Introduction: Why Diet Matters in the Fight Against Cancer
Diet is one of the most controllable factors in cancer prevention and treatment — yet it remains consistently underutilized. It functions both as a modifiable risk factor for chemoprevention and as a meaningful support strategy for patients already managing a diagnosis.
The evidence is substantial. According to the Global Burden of Disease Study 2019, 44.4% of global cancer deaths and 42.0% of cancer disability-adjusted life years (DALYs) are attributable to estimated risk factors, many of which are dietary and lifestyle-related. This means nearly half of all cancer mortality is tied to factors within patient control.
That figure reframes how patients and practitioners approach the disease. Diet isn't a passive afterthought — it actively shapes cancer biology through inflammation, oxidative stress, epigenetics, and metabolic signaling.
This article examines specific plant compounds with demonstrated chemopreventive potential, metabolic dietary strategies that exploit cancer cell vulnerabilities, and how dietary patterns can work alongside conventional treatment. Diet is not a replacement for medical care, but it is a lever patients can pull — whether the goal is risk reduction or supporting recovery.
How Diet Shapes Cancer Risk: The Evidence Base
Carcinogenesis and Dietary Intervention Points
Cancer develops through a multistep process—initiation, promotion, and progression—and dietary factors can intervene at each stage. Epidemiological evidence consistently links specific dietary patterns to cancer incidence across populations. Diet is one of the few major cancer risk factors that is directly modifiable.
Foods Linked to Increased Cancer Risk
The evidence against certain dietary habits is clear:
- Processed and red meats: The International Agency for Research on Cancer concluded that each 50-gram portion of processed meat eaten daily increases colorectal cancer risk by 18%
- Alcohol consumption: A comprehensive meta-analysis found that heavy drinkers compared with non-drinkers showed relative risks of 5.13 for oral/pharyngeal cancer, 4.95 for esophageal squamous cell carcinoma, and 2.65 for laryngeal cancer
- High-sugar and refined carbohydrates: Diets promoting obesity and insulin resistance create pro-inflammatory, pro-growth environments favorable to cancer development

Protective Dietary Patterns
Diets rich in whole plant foods—fruits, vegetables, whole grains, and legumes—associate with lower cancer risk, particularly for colorectal and breast cancers. These foods provide antioxidants, anti-inflammatory compounds, and fiber that modulate cancer-promoting pathways. The protective effect appears strongest when these foods displace, rather than simply supplement, processed and animal-derived foods.
The Dietary Pattern Paradigm
Major authoritative bodies have shifted from nutrient-centric approaches to emphasizing whole dietary patterns. The American Cancer Society explicitly states: "One major change in cancer prevention guidelines over time has been a shift from a reductionist or nutrient-centric approach to a more holistic concept of diet that is characterised as dietary patterns."
That shift matters in practice: it means the combination and context of foods—not individual nutrients in isolation—drives cancer risk up or down.
Understanding Dietary Chemoprevention
Dietary chemoprevention uses food components to actively block or reverse cancer development at different stages:
- Primary chemoprevention: Preventing cancer in healthy or at-risk populations
- Secondary chemoprevention: Preventing progression from premalignant lesions
- Tertiary chemoprevention: Preventing recurrence after treatment
Each stage targets distinct biological windows—meaning dietary intervention remains relevant whether someone is managing risk, a premalignant diagnosis, or life after cancer care.
Phytochemicals and Plant Compounds With Chemopreventive Properties
Phytochemicals are bioactive plant compounds—polyphenols, carotenoids, alkaloids, isothiocyanates—that interact with cancer-relevant signalling pathways. Their general mechanisms include affecting cellular stress pathways being investigated in nutritional oncology research, inhibiting cell proliferation, modulating inflammation (NF-κB, COX-2), and enhancing immune surveillance.
Curcumin: Promising Biology, Limited Bioavailability
Curcumin from turmeric is one of the most studied phytochemicals in oncology. Phase II trials have tested it in pancreatic cancer and familial adenomatous polyposis (FAP), showing polyp reduction in FAP patients when combined with quercetin.
The bioavailability problem: A Phase II trial in advanced pancreatic cancer using 8g/day found peak plasma levels of only 22-41 ng/mL, indicating extremely poor oral absorption. Despite this limitation, one patient experienced a brief but marked 73% tumor regression.
Enhanced delivery systems: To overcome poor absorption, micellar and colloidal formulations have been developed. A human crossover study demonstrated that micellar formulations showed 136-fold higher AUC levels than standard extract, making therapeutic systemic exposure achievable.
| Formulation | Bioavailability Impact | Clinical Context |
|---|---|---|
| Standard extract | Peak 22-41 ng/mL at 8g/day | Poor systemic efficacy |
| Micellar curcumin | 136-fold higher AUC | Achieves therapeutic levels |
| Dried colloidal | 72.9-fold higher AUC | High unconjugated absorption |
Resveratrol and Tea Polyphenols (EGCG)
Resveratrol from red grapes and berries has been investigated for its effects on cellular stress pathways across multiple cancer cell lines and modulates immune surveillance via natural killer cell activation. Key findings include:
- A 4-month trial in middle-aged men found the highest dose lowered dehydroepiandrosterone-sulphate (DHEAS) by 50%, suggesting hormone-modulating effects relevant to prostate cancer.
EGCG from green tea reduces oxidative DNA damage. Clinical evidence shows:
- A randomized trial in heavy smokers showed a 31% decrease in urinary 8-OHdG after 4 months of decaffeinated green tea, a biomarker of oxidative stress and DNA damage.
- In a Phase II trial for high-risk oral premalignant lesions, 50% of patients receiving green tea extract showed clinical response versus 18.2% on placebo.
Cruciferous Vegetables and Isothiocyanates
Sulforaphane and other isothiocyanates from broccoli, cauliflower, and watercress show strong chemopreventive data.
A prospective study in Shanghai found individuals with detectable isothiocyanates in urine had a 35% reduced risk of lung cancer (relative risk 0.65). For bladder cancer, higher consumption of raw broccoli was associated with greater than two-fold reduction in disease-specific death (hazard ratio 0.43).
Beyond plant-derived compounds, certain dietary fats carry equally compelling chemopreventive evidence — particularly the marine omega-3 fatty acids.
Omega-3 Fatty Acids
Marine omega-3s—EPA and DHA—exert anti-inflammatory effects by inhibiting arachidonic acid-derived eicosanoids, reducing COX-2 overexpression, and supporting anti-inflammatory gut microbiota.
A meta-analysis of 25 prospective studies found highest fish consumption associated with 6% reduced colorectal cancer risk. In a randomized trial, FAP patients taking 2g/day of highly purified EPA reduced rectal polyp number by 22.4% and polyp size by 29.8% over 6 months — results comparable to drug-based interventions in this population.

Metabolic Dietary Strategies That Target Cancer Cells
Cancer cells exhibit distinct metabolic vulnerabilities not shared by healthy cells, including dependence on specific amino acids and altered glucose metabolism (the Warburg effect). Dietary manipulation can selectively pressure cancer cells while sparing normal tissue.
Three strategies have the strongest research foundation: methionine restriction, ketogenic diets, and fasting-mimicking diets. Each targets a different vulnerability — and each carries important caveats.
Methionine Restriction: Exploiting the Hoffman Effect
Many cancer cells are auxotrophic for methionine — unable to survive when methionine is replaced with its precursor homocysteine, while normal cells adapt normally. This metabolic vulnerability is called the Hoffman Effect.
Restricting dietary methionine through a plant-dominant, low-methionine diet can selectively stress cancer cells. NORI has developed structured protocols based on this principle, applied in case reports of Stage IV cancers including thyroid and breast cancer.
These protocols typically limit methionine and cysteine intake to no more than 10 mg/kg body weight per day through predominantly fruit and vegetable-based diets, with total protein restricted to less than 0.3 grams per kilogram daily.
A Phase I/II trial combining methionine deprivation with chemotherapy for melanoma and glioma achieved 41-53% plasma methionine reduction but produced no complete or partial responses. Biochemical effects are achievable — but translating them into measurable clinical responses requires further optimization, likely through combination approaches.
Ketogenic and Low-Glucose Dietary Approaches
Ketogenic diets (high fat, very low carbohydrate) reduce circulating glucose and insulin, potentially limiting fuel supply for glycolytic cancer cells. The theoretical rationale is solid; the clinical evidence is more complicated.
A systematic review found ketogenic diets significantly reduced fat mass, visceral fat, insulin levels, blood glucose, fatigue, and insomnia, but survival benefits remain unproven.
A critical safety concern tempers enthusiasm: recent preclinical data shows that while ketogenic diets suppress primary breast tumors, they actively promote lung metastasis via BACH1-mediated transcription. This finding raises serious questions about continuous ketogenic diet use in certain solid tumors.
Caloric Restriction and Fasting-Mimicking Diets
Of the three strategies, fasting-mimicking diets (FMDs) currently carry the strongest clinical evidence for cancer patients. A randomized trial of a 5-day FMD effectively reduced body weight, waist circumference, systolic blood pressure, and IGF-1, demonstrating safe modulation of key growth factors.
In the DIRECT Phase II trial for HER2-negative breast cancer, FMD improved pathological response rates — indicating 90-100% tumor cell loss — when used alongside neoadjuvant chemotherapy, making it the most clinically validated metabolic adjunct currently supported by randomized trial data.
| Strategy | Mechanism | Clinical Evidence |
|---|---|---|
| Methionine restriction | Exploits cancer cells' inability to survive on homocysteine alone | 41-53% plasma reduction; no objective responses in early trials |
| Ketogenic diet | Reduces glucose/insulin | Lowers IGF-1/glucose but promotes metastasis in some breast cancer models |
| Fasting-mimicking diet | Reduces IGF-1/glucose; protects normal cells | Improves pathological response when combined with chemotherapy |

Important: These metabolic strategies require medical supervision. Cancer patients' nutritional needs differ significantly from healthy individuals — personalized, evidence-informed programs are appropriate, not self-directed extreme diets.
Dietary Support During Conventional Cancer Treatment
The Malnutrition Crisis in Cancer Care
Cancer treatments—chemotherapy, radiation, immunotherapy, surgery—directly compromise nutrition through nausea, taste changes, mouth sores, appetite loss, and malabsorption. A comprehensive meta-analysis of 65 studies involving 31,330 patients found overall malnutrition prevalence of 41%.
Malnutrition drives treatment toxicity: In metastatic breast cancer patients receiving chemotherapy, 50% of sarcopenic patients experienced severe toxicity compared to only 20% of non-sarcopenic patients. Sarcopenia was the only significant predictor of toxicity even after controlling for performance status and albumin.
Shifting Nutritional Priorities During Treatment
Cancer patients often need more protein and calories during active treatment—not less—to prevent muscle wasting and cachexia. This fundamentally changes what "healthy eating" looks like compared to prevention-focused diets.
ESPEN guidelines recommend:
- Energy intake: 25-30 kcal/kg/day
- Protein intake: Above 1 g/kg/day, ideally up to 1.5 g/kg/day
This contrasts sharply with metabolic restriction strategies appropriate only under medical supervision.
Supplement-Treatment Interactions
Beyond macronutrients, supplement choices matter too. While some phytochemicals may sensitize cancer cells to chemotherapy or radiation, others can work against treatment goals. Key interaction risks include:
- High-dose antioxidants (vitamins C, E, beta-carotene): may reduce oxidative damage that chemotherapy depends on
- Herbal supplements (St. John's Wort, turmeric at therapeutic doses): can affect drug metabolism via liver enzymes
- Fat-soluble vitamins at megadoses: may accumulate and interact with radiation protocols
Working with a registered dietitian or nutritional oncology specialist ensures dietary choices are coordinated with the oncology team, maximizing benefit while minimizing risk.
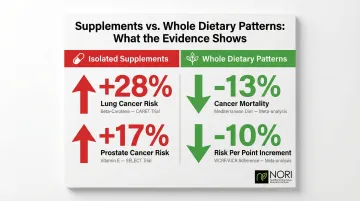
Dietary Patterns vs. Isolated Supplements: What the Evidence Shows
The Danger of Isolated High-Dose Supplements
Large-scale randomised trials definitively prove that isolated, high-dose antioxidant supplements can cause active harm:
- Beta-carotene: The CARET trial in smokers was halted early because the active-treatment group had 28% increased lung cancer risk (relative risk 1.28)
- Vitamin E: The SELECT trial found vitamin E increased prostate cancer risk by 17% in healthy men (hazard ratio 1.17)
Both trials show what happens when single nutrients are pulled out of the whole foods they naturally occur in — the protective effects disappear, and harm can follow.
Whole Dietary Patterns: Consistent Protective Benefits
Where isolated supplements have repeatedly failed, whole-food dietary patterns show consistent cancer risk reduction:
- Mediterranean diet: An updated meta-analysis of 117 studies found highest adherence associated with 13% reduced cancer mortality (relative risk 0.87)
- WCRF/AICR guidelines: A continuous meta-analysis showed cancer risk reduced by 10% for each 1-point increment in adherence score
| Intervention | Trial/Pattern | Outcome |
|---|---|---|
| Beta-carotene supplement | CARET | 28% INCREASE in lung cancer |
| Vitamin E supplement | SELECT | 17% INCREASE in prostate cancer |
| Mediterranean diet | Meta-analysis | 13% DECREASE in cancer mortality |
| WCRF/AICR adherence | Meta-analysis | 10% DECREASE per point increment |
When Supplements May Be Appropriate
Whole-food dietary patterns form the foundation. Targeted supplementation has a role in specific situations:
- Correcting documented deficiencies (vitamin D, omega-3s)
- Specific clinical contexts under medical guidance
- Enhanced delivery formulations of proven compounds (micellar curcumin)
Avoid high-dose isolated antioxidants without medical supervision — especially during active cancer care.
Frequently Asked Questions
What is cancer chemoprevention and how does diet fit into it?
Chemoprevention uses natural or synthetic agents—including dietary compounds—to inhibit or reverse cancer development at the initiation, promotion, or progression stages. Diet is one of the most accessible chemopreventive tools available, capable of modulating inflammation, oxidative stress, hormone signaling, and immune function across all risk levels.
Which foods have the strongest scientific evidence for reducing cancer risk?
Cruciferous vegetables (broccoli, cauliflower), berries, tomatoes, green tea, legumes, and whole grains show the strongest evidence. Dietary variety matters more than any single "superfood"—the synergistic effects of diverse plant compounds consistently outperform isolated foods.
Can diet alone support cancer recovery?
Diet alone is not a proven standalone cancer care approach. However, targeted dietary strategies can create conditions unfavorable for cancer cell growth, support conventional cancer care outcomes, and improve quality of life. Rigorous nutritional protocols work best when integrated with medical oversight and conventional care, not as replacements.
What is methionine restriction and why is it relevant to cancer?
Many cancer cells cannot metabolize methionine precursors like homocysteine—a metabolic vulnerability called the Hoffman Effect. Low-methionine diets (achieved through plant-based eating) selectively deprive cancer cells of this essential amino acid while healthy cells adapt normally.
Are dietary supplements helpful or harmful for cancer patients?
The evidence is highly context-dependent. Some supplements show clear benefit—omega-3s, vitamin D, and enhanced curcumin formulations in specific contexts—while others prove harmful at high doses (beta-carotene in smokers, high-dose vitamin E during active treatment). Individual assessment with a qualified provider is essential.
How does diet interact with chemotherapy and other cancer care?
Certain phytochemicals can sensitize cancer cells to chemotherapy or help mitigate side effects, while high-dose antioxidant supplements taken concurrently may reduce treatment efficacy by protecting cancer cells from oxidative damage. Informed, supervised dietary choices during active treatment are critical—never self-prescribe supplements during chemotherapy without oncologist approval.
Key Takeaway
Diet is one of the most powerful modifiable factors in both cancer prevention and treatment support. The strongest evidence favors whole dietary patterns—particularly plant-forward eating rich in diverse phytochemicals—over isolated supplements. Metabolic strategies targeting cancer cell vulnerabilities show real promise but require medical supervision and continued clinical validation.
For patients seeking to integrate nutritional strategies with their care plan, working with specialists in nutritional oncology—such as the consultation services offered by NORI—ensures approaches that are evidence-based, personalized, and designed to support rather than interfere with treatment outcomes.


