
Introduction
Dietary supplements are remarkably popular among cancer patients and survivors — research shows that up to 70.4% use supplements, compared to 51.2% of individuals without cancer. Yet despite this widespread use, a troubling disclosure gap exists: between 31% and 68% of cancer patients who take supplements never tell their oncology team about them.
This silence creates genuine danger. Many cancer patients turn to supplements hoping to boost recovery, strengthen immunity, or counter treatment side effects. But the safety landscape for this population is vastly more complex than marketing claims suggest.
Several specific risks stand out:
- Some supplements protect cancer cells from oxidative damage — the very mechanism chemotherapy and radiation rely on to kill tumors
- Others accelerate drug metabolism in the liver, reducing treatment effectiveness
- Still others increase surgical bleeding risk or interact dangerously with anesthesia
This guide covers the full picture. You'll learn which supplements carry real risks during active treatment, what interactions to watch for, and how to make safer decisions in consultation with your care team.
TL;DR
- Dietary supplements reach the market without FDA pre-approval for safety or effectiveness — cancer patients carry the greatest risk
- High-dose antioxidants, beta-carotene, Vitamin E, and selenium have increased cancer risk in major clinical trials
- Supplements can reduce chemotherapy effectiveness, worsen radiation side effects, or increase surgical bleeding risk
- "Natural" doesn't mean safe — high-dose supplements are among the most common causes of treatment complications
- Tell your oncology team about every supplement you take, before and during treatment — no exceptions
Why Supplement Safety Is More Complex for Cancer Patients
Cancer patients face risks the general population does not. Supplements can interact with cancer drugs, radiation, and cancer cell biology in ways that compound harm well beyond any standalone risk — often in directions that are neither obvious nor labeled.
The FDA Regulatory Gap
The FDA does not require dietary supplements to be tested or approved before they reach the market. Manufacturers must report serious adverse events after the fact, but compliance is inconsistent. This regulatory gap leaves cancer patients — whose bodies and immune systems are often compromised — especially vulnerable.
According to the FDA, supplements are regulated under the Dietary Supplement Health and Education Act (DSHEA), which allows structure/function claims like "supports immune health" or "promotes cellular repair" without FDA review. These claims can easily be misread as disease prevention or treatment claims by patients hoping for options.
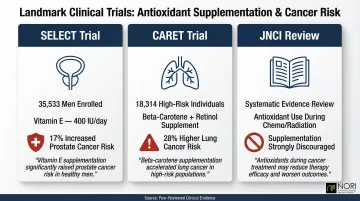
Major Clinical Trials Show Harm, Not Benefit
Three landmark trials challenged long-held assumptions about supplement safety:
- SELECT Trial (Prostate Cancer): In a study of 35,533 men, those taking 400 IU of Vitamin E daily developed prostate cancer at significantly higher rates than the placebo group (hazard ratio 1.17), representing an absolute increase of 1.6 cases per 1,000 person-years
- CARET Trial (Lung Cancer): Among 18,314 high-risk individuals (smokers and asbestos-exposed workers), those taking 30 mg beta-carotene and 25,000 IU retinol daily had a 28% higher lung cancer risk and 17% higher overall mortality than placebo
- Journal of the National Cancer Institute (JNCI) Review: A 2008 analysis concluded that supplemental antioxidants during chemotherapy and radiation should be discouraged due to tumor protection potential
The Nutrient Status Factor
High-dose supplementation is most likely to cause harm in people who already have adequate nutrient levels — which most Western cancer patients do. A 2014 case-cohort study of the SELECT trial found that selenium supplementation increased high-grade prostate cancer risk by 91% in men with high baseline selenium status.
The assumption that "more is better" is biologically unsound and, for cancer patients, clinically dangerous.
Misleading Labeling
Supplement labels can mislead vulnerable patients. Structure/function claims aren't reviewed by the FDA, and a 2018 study in JAMA Network Open identified 776 adulterated dietary supplements in the FDA's Tainted Supplements database — 97.6% contained unapproved pharmaceutical ingredients not declared on the label.
Supplements and Cancer Treatment: Key Safety Risks
The risks aren't theoretical. They're dose-dependent, treatment-specific, and well-documented in oncology literature. The three highest-risk contexts are active chemotherapy, radiation therapy, and the surgical window.
During Chemotherapy
High-Dose Antioxidants: The ROS Interference Problem
Many chemotherapy drugs work by generating reactive oxygen species (ROS) that damage cancer cell DNA and affect cellular processes being investigated in research. High-dose antioxidant supplements — Vitamins C and E, coenzyme Q10, selenium — may counteract this by neutralizing the same oxidative damage chemo depends on.
The NCI notes that insufficient information exists to determine if antioxidants are safe during treatment, as they may protect cancer cells alongside healthy ones. Clinical evidence supports this concern: the SWOG S0221 trial found that breast cancer patients using antioxidant supplements both before and during chemotherapy had a 41% increased hazard of recurrence.
St. John's Wort: A Documented Drug Interaction
St. John's Wort is one of the most dangerous supplements during chemotherapy. It induces liver enzymes (CYP3A4 and P-glycoprotein) that speed up drug metabolism, reducing effective drug concentrations in the bloodstream.
| Chemotherapy Drug | Effect of St. John's Wort | Clinical Impact |
|---|---|---|
| Irinotecan | Decreases active metabolite (SN-38) by 42% | Worsens myelosuppression, reduces efficacy |
| Imatinib | Increases clearance by 43%, decreases AUC by 30% | Subtherapeutic drug levels, treatment failure |
| Docetaxel | Decreases AUC from 3,035 to 2,682 ng·h/mL | Reduced chemotherapy effectiveness |

During Radiation Therapy
High-Dose Antioxidants: The Same Mechanism, A Different Treatment
Radiation therapy kills cancer cells through oxidative damage to DNA — the same mechanism that antioxidants interfere with during chemotherapy. The American Society for Radiation Oncology (ASTRO) advises patients to avoid high-dose antioxidants during radiation therapy for this reason.
A randomized trial of 540 head and neck cancer patients found that supplementation with high doses of alpha-tocopherol (400 IU/d) and beta-carotene (30 mg/d) during radiation tended to increase local tumor recurrence (HR 1.37), suggesting antioxidants might compromise radiation efficacy.
Photosensitivity and Skin Reactions
St. John's Wort contains hypericin, a known photosensitizing agent. Clinical case reports document radiation recall dermatitis occurring one year after radiotherapy, triggered by St. John's Wort ingestion. Some supplements increase skin photosensitivity, worsening radiation dermatitis and acute skin reactions.
Before or After Surgery
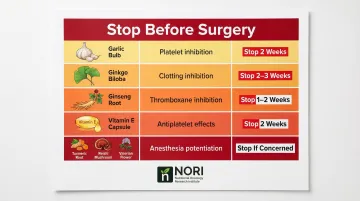
Standard Cessation Window: 2 Weeks
Surgery introduces a third risk category: bleeding and anesthesia interactions. The Society for Perioperative Assessment and Quality Improvement (SPAQI) and the American Society of Anesthesiologists (ASA) recommend discontinuing most dietary supplements 2 weeks prior to surgery for this reason.
| Supplement | Perioperative Risk | Recommended Cessation |
|---|---|---|
| Garlic, Ginkgo, Ginseng | Inhibits platelet aggregation; increases bleeding | Hold 2 weeks before surgery |
| Vitamin E (>400 IU/day) | Inhibits platelets; antagonizes vitamin K-dependent clotting | Hold 2-3 weeks before surgery |
| Turmeric/Curcumin | Inhibits thromboxane metabolism; affects clotting | Hold 2 weeks before surgery |
| Medicinal Mushrooms | Prolongs clotting times; antiplatelet effects | Hold 1-2 weeks before surgery |
| Valerian Root | Potentiates anesthesia sedation via GABA receptors | Stop if concerned about sedation |
Post-Surgical Healing Concerns
Risks don't end once surgery is complete. Key concerns in the recovery window include:
- Vitamin E (high doses) — impairs wound healing by inhibiting collagen cross-linking
- Fish oil — may interact with post-operative anticoagulants such as warfarin, increasing bleed risk
- St. John's Wort — accelerates metabolism of opioid pain medications, potentially reducing their effectiveness
- High-dose zinc — in excess, may suppress immune recovery during the healing period
Always review all active supplements with your surgical team before and after the procedure.
Safety Guidelines When Considering Supplements During Cancer Care
"Avoid all supplements" isn't always realistic or appropriate guidance. Oncologists sometimes prescribe Vitamin D or calcium for bone protection during hormone therapy, and genuine nutritional deficiencies do occur. The goal is use that is informed, disclosed, and supervised by your care team.
Before Starting Any Supplement
Many patients don't tell their doctors — and that's the single most dangerous practice. Research from Fred Hutch shows that 46.1% of cancer survivors use dietary supplements on their own without consulting healthcare providers.
Before taking any supplement, ask your oncologist or registered oncology dietitian:
- Does this interact with my current treatment?
- What dose is safe at my current nutritional status?
- Is there a whole-food source that provides this nutrient more safely?
Nutrient deficiencies should be confirmed through bloodwork before supplementing. Supplementing when no deficiency exists carries risk without benefit — blood tests guide appropriate dosing and prevent unnecessary exposure.
Once you've confirmed a deficiency and gotten clearance from your care team, the next step is choosing a product you can actually trust.
Choosing Quality Supplements (When Cleared by Your Care Team)
Because the FDA doesn't pre-approve supplements, third-party verification is the only reliable method to confirm product purity and avoid harmful contaminants.
| Verification Program | What It Tests | How to Identify |
|---|---|---|
| USP (US Pharmacopeia) | Ingredient accuracy, potency, contaminants (heavy metals, microbes, pesticides), cGMP manufacturing | Circular logo with "USP Dietary Supplement Verified" |
| NSF International | Composition accuracy, contaminant testing (lead, mercury, arsenic, pesticides), annual facility audits | "NSF Contents Tested & Certified" mark |
| ConsumerLab.com | Potency, safety (contaminants), performance (USP standards for disintegration/dissolution) | "CL Seal of Approval" on packaging |
When your care team approves specific nutraceutical use, look for products formulated with cancer-specific safety in mind. Oncology-focused nutraceuticals are developed with awareness of treatment interactions and cancer biology — a meaningful distinction from general wellness supplements designed without that clinical context.
Common Supplement Mistakes Cancer Patients Should Avoid
Mistake: Assuming "Natural" Means Safe
Many natural compounds are biologically active in ways that directly affect cancer cell behavior and drug metabolism:
- Laetrile (Vitamin B17): Converts to cyanide in the body; clinical trials found no cancer benefit but documented severe cyanide poisoning (tachycardia, altered mental status, lactic acidosis, coma)
- Comfrey: Contains pyrrolizidine alkaloids that cause hepatic sinusoidal obstruction syndrome (veno-occlusive disease) and severe liver injury
- Kava: Linked to clinically apparent acute liver injury, which can be severe and fatal
Mistake: Self-Prescribing High-Dose "Anti-Cancer" Vitamins
The same clinical trials that drove supplement popularity (early observational studies suggesting beta-carotene or selenium were protective) later produced RCT evidence of harm. Nutritional oncology evidence evolves rapidly, and findings from observational studies are frequently overturned — or complicated — by later randomized trials. Interpreting this research accurately requires clinical context, not just access to studies.
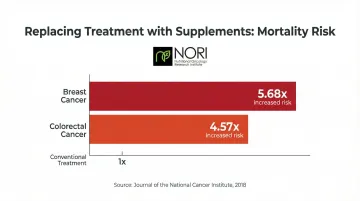
Mistake: Replacing an Active Treatment Plan Without Expert Guidance
That same complexity is why abandoning a working treatment plan based on unvetted supplement protocols carries real risk. A 2018 study in the Journal of the National Cancer Institute found that patients who refused conventional cancer care in favor of alternative medicine alone had a significantly greater risk of death:
- Breast cancer: 5.68-fold increased risk of death
- Colorectal cancer: 4.57-fold increased risk of death
Nutritional support and nutraceutical protocols can be powerful additions to a patient's overall strategy. The risk lies in self-directing major treatment changes — dropping therapies that are working, or delaying those that are needed — based on supplement marketing rather than expert clinical assessment.
Conclusion
Supplement safety for cancer patients depends on treatment type, dosage, individual nutritional status, and cancer biology — no universal answer applies across the board. That makes active communication with a qualified care team non-negotiable; self-prescribing based on independent research carries real risk.
That said, nutritional medicine is a legitimate and valuable complement to treatment when approached with the same rigor as any other medical decision. That means:
- Evidence-based: grounded in research, not marketing claims
- Disclosed: every supplement shared with the oncology team upfront
- Supervised: dosing and timing guided by qualified practitioners
- Tailored: matched to the specific cancer type, stage, and treatment protocol
NORI's home-based nutritional support programs are built around exactly this framework — providing science-driven guidance that works alongside, not in place of, a patient's existing care plan.
Frequently Asked Questions
Which vitamin is called health-supporting?
No single vitamin has been proven to support cancer wellness or provide cancer care in clinical trials. The VITAL trial found that 2,000 IU/day of vitamin D3 did not reduce invasive cancer incidence after 5.3 years. Some high-dose antioxidants have actually increased cancer risk in certain populations.
Can supplements interfere with chemotherapy or radiation?
Yes. High-dose antioxidants, St. John's Wort, and certain herbal products are documented to reduce treatment effectiveness. They can shield cancer cells from oxidative damage or speed up drug metabolism, lowering effective drug concentrations in the bloodstream.
Are antioxidant supplements safe to take during cancer care?
Most oncology organizations advise against high-dose antioxidant supplementation during active cancer care. Oxidative damage is part of how chemo and radiation kill cancer cells — antioxidants may counteract this mechanism. Low-dose antioxidants from food sources are generally not a concern.
What supplements should cancer patients avoid?
Discuss these categories with your oncologist before use:
- High-dose antioxidants (Vitamins A, C, E, beta-carotene) during active treatment
- St. John's Wort, which interacts with many cancer drugs
- Blood-thinning supplements (fish oil, turmeric, garlic, melatonin) around surgery
- Any product marketed as an alternative cancer cure
Is it safe to take vitamins while on cancer medication?
The answer depends on which vitamin, what dose, and which cancer drug is involved. Some vitamins (like Vitamin D prescribed for bone health on hormone therapy) are appropriate, while others at high doses can interact with cancer drugs. Always tell your oncology team about every supplement, including dose and frequency.
Should cancer patients take supplements during treatment or only after?
Most oncologists advise limiting non-prescribed supplements during active treatment. That said, specific supplements — Vitamin D, calcium, oral glutamine — may be recommended to manage side effects. After treatment, a care team should still guide any supplement use to avoid promoting recurrence.


