
Introduction
Colon cancer is uniquely sensitive to nutritional intervention. Research estimates dietary and nutritional factors account for approximately one-third of the global colorectal cancer burden — the highest diet-related burden among all cancer types. Because the colon is directly exposed to everything you consume, supplementation strategies carry more weight here than in almost any other cancer type.
Standard treatments — surgery, chemotherapy, and radiation — can significantly deplete key nutrients, impair absorption, and disrupt the gut microbiome. That creates a genuine need for targeted nutritional support, not general wellness supplementation.
This guide focuses specifically on supplements for colon cancer patients — not just prevention — covering what supports recovery, reduces inflammation, and complements treatment. All supplementation decisions should be discussed with your healthcare provider before implementation.
TLDR
- Vitamin D and calcium are among the most researched supplements for colon cancer, with adequate levels linked to better survival outcomes
- Folate, curcumin, and omega-3 fatty acids each have clinical research behind them — dosing and timing significantly affect their benefit
- Probiotics and dietary fiber support gut microbiome health, which influences colorectal cancer progression and how well patients respond to treatment
- High-dose antioxidant supplements may interfere with chemotherapy effectiveness; medical oversight is essential
- A plant-forward, high-fiber diet combined with methionine restriction is an evidence-backed approach shown to limit cancer cell proliferation
Why Nutrition Matters for Colon Cancer Patients
Colon cancer stands apart from other cancers in how directly nutrition influences outcomes. According to the Global Burden of Disease Study 2021, the global age-standardized mortality rate of colorectal cancer attributable to total dietary risks was 4.8 per 100,000 population. The colon's constant exposure to dietary components means what you eat—and supplement—can either support or hinder recovery.
Standard treatments create specific nutritional challenges:
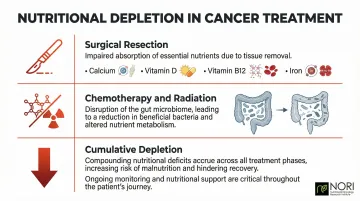
- Surgical resections (colectomy, ileostomy) impair absorption of calcium, vitamin D, vitamin B12, and iron
- Chemotherapy and radiation disrupt the gut microbiome, reducing beneficial bacteria while expanding potentially harmful species like Enterococcus
- Cumulative nutrient depletion across treatment phases compounds these deficits over time
Given these treatment-driven deficits, supplements for colon cancer patients serve different purposes than prevention supplements. The goal shifts to managing side effects, supporting immune function, reducing cancer cell proliferation, and maintaining quality of life — and the evidence supporting each use case varies considerably across these goals.
Best Supplements for Colon Cancer Patients
The supplements below are selected based on clinical and observational research specifically relevant to colorectal cancer patients. Dosages should always be personalized with medical guidance.
Vitamin D
Vitamin D is one of the most studied nutrients in colorectal cancer. Adequate blood levels of 25-hydroxyvitamin D are associated with lower risk of developing colorectal cancer and, importantly, with better survival rates in diagnosed patients.
The mechanism: Vitamin D has been studied for its effects on cellular health, affecting cellular pathways, as investigated in nutritional oncology research, and supports immune modulation. A meta-analysis of 17 studies involving 17,770 colon cancer patients found that the highest versus lowest categories of circulating 25(OH)D were associated with a hazard ratio of 0.64 for overall survival and 0.65 for colon cancer-specific survival.
Many colon cancer patients are deficient in Vitamin D, especially those with limited sun exposure or impaired gut absorption. In the CALGB/SWOG 80405 trial of 1,043 patients with metastatic colon cancer, median overall survival was 32.6 months for those with the highest baseline Vitamin D levels compared to 24.5 months for those with the lowest levels.
Dosing considerations: The NIH recommends 600 IU daily for adults up to age 70, and 800 IU for adults 71 and older. The phase II SUNSHINE trial studied high-dose Vitamin D3 (8,000 IU loading dose, then 4,000 IU daily) and showed a promising hazard ratio of 0.64 for progression-free survival. However, the subsequent phase III SOLARIS trial found no benefit in untreated metastatic colon cancer at these doses.
Test your baseline 25(OH)D levels first and supplement to correct deficiencies — ultra-high doses without medical supervision carry risk and have not shown consistent benefit.
Calcium
Calcium complements Vitamin D and has its own protective mechanism. In the colon, calcium binds to bile acids and fatty acids, forming insoluble soaps that reduce mucosal cell exposure to carcinogens.
Meta-analyses suggest that each 300 mg/day increase in supplementary calcium is associated with approximately 9% reduced risk of colon cancer. The protective effect appears most relevant for adenoma recurrence—a key concern for colon cancer patients and survivors.
The landmark Calcium Polyp Prevention Study found that 1,200 mg/day of elemental calcium reduced the adjusted risk ratio for any adenoma recurrence to 0.85 compared to placebo.
Important caution: Excessive calcium supplementation can have downsides. The evidence points to benefit at moderate intakes around 700–800 mg/day from diet and supplements combined, not megadosing.
Folate (Vitamin B9)
Folate plays a critical role in DNA synthesis, repair, and methylation. Deficiency increases genomic instability and colorectal cancer risk. Observational studies consistently link higher folate intake to reduced risk of colorectal adenomas and cancer, particularly in people with low baseline folate status.
One critical nuance: supplemental folic acid is NOT the same as dietary folate for all patients. A landmark JAMA trial revealed that high-dose synthetic folic acid (1 mg/day) did not prevent adenomas and actually increased the risk of advanced lesions (RR 1.67) and having ≥3 adenomas in patients with prior polyp history.
Best food sources include:
- Lentils: 358 mcg per cooked cup
- Spinach: 263 mcg per cooked cup
- Black beans: 256 mcg per cooked cup
- Broccoli: 103 mcg per cooked cup
Patients with inflammatory bowel disease or malabsorption conditions may benefit from supervised folate supplementation. Those with a history of colon cancer or adenomas should strictly avoid high-dose synthetic folic acid and instead source protective natural folate from food.
Curcumin (from Turmeric)
Curcumin is one of the most studied natural compounds in colorectal cancer research. It demonstrates anti-inflammatory, antioxidant, and wellness-supporting properties through multiple mechanisms—inhibiting colon cancer cell invasion, suppressing inflammatory pathways (NF-κB), and down-regulating COX-2 expression.
A pilot study in familial adenomatous polyposis patients showed that 480 mg curcumin combined with 20 mg quercetin three times daily for six months reduced polyp number by 60.4% and polyp size by 50.9%.
Bioavailability is the central challenge with curcumin. Oral doses as high as 10-12 grams yield only trace plasma concentrations, and a 2018 trial using 3,000 mg/day of pure curcumin failed to reduce adenoma burden in FAP patients for exactly this reason.
Solution: Look for bioavailability-enhanced formulations:
- Piperine combination: Co-administration of 20 mg piperine with 2 g curcumin increased bioavailability by 2,000% in healthy volunteers
- Nanoparticle formulations: Theracurmin demonstrated a 27-fold higher absorption compared to standard curcumin powder

Curcumin works best as a complementary support agent alongside other interventions. Larger randomized trials in colon cancer patients remain limited, so formulation choice and dosing should be guided by a knowledgeable practitioner.
Omega-3 Fatty Acids (Fish Oil)
Omega-3 polyunsaturated fatty acids (EPA and DHA from fatty fish or fish oil supplements) have demonstrated anti-inflammatory properties and are being studied as adjuncts to colorectal cancer chemotherapy.
Data from the Nurses' Health Study shows striking results: colon cancer patients who increased their marine omega-3 intake by just 0.15 g/day post-diagnosis had a hazard ratio of 0.30 for colon cancer-specific mortality—representing a 70% reduction compared to those who did not change their intake.
The mechanisms include COX-2 inhibition and modulation of the tumor microenvironment. Omega-3 fatty acids are precursors to Specialized Pro-resolving Mediators (SPMs) like Resolvins. These compounds stimulate macrophage clearance of tumor debris generated by chemotherapy and suppress pro-inflammatory cytokines like IL-6 and TNFα.
Key findings from clinical research:
- The EMT2 trial is evaluating 4 g/day of EPA on colon cancer recurrence after surgery for resectable liver metastases
- Earlier EMT phase 2 trials showed 2 g/day EPA was safe, incorporated into tumor tissue, and associated with reduced tumor vascularity
Note that the evidence for standalone omega-3 supplementation in colorectal cancer prevention is inconsistent. The clearest benefit appears as anti-inflammatory support and as an adjunct during active treatment.
Probiotics and Prebiotic Fiber
The colon hosts trillions of bacteria, and imbalances in gut microbiota are closely linked to colorectal cancer development. Dysbiosis—characterized by enrichment of harmful bacteria like Fusobacterium nucleatum and polyketide synthase-positive E. coli—promotes tumor progression and treatment resistance.
Synbiotics (prebiotics + probiotics combined) show measurable benefits: A 12-week randomized trial in 37 colon cancer patients and 43 polypectomized patients used a synbiotic containing oligofructose-enriched inulin plus Lactobacillus rhamnosus GG and Bifidobacterium lactis Bb12. Results showed significantly reduced colorectal proliferation, decreased necrosis-inducing activity in fecal water, and less DNA damage in colonic mucosa.
Probiotics and prebiotics promote production of short-chain fatty acids like butyrate, which fuels colonocytes and exerts anti-inflammatory effects throughout the gut.
Critical safety warning: While synbiotics support gut health, Memorial Sloan Kettering Cancer Center warns that patients who are immunocompromised, critically ill, or have a central venous catheter should NOT take live probiotics due to the risk of bacteremia and systemic infections. Patients on immunosuppressive chemotherapy regimens must consult their oncologist before starting probiotics.
Foods to Eat and Avoid During Colon Cancer
Foods to Prioritize
A plant-based, high-fiber diet provides the strongest nutritional foundation for colon cancer patients:
- Cruciferous vegetables (broccoli, kale, Brussels sprouts, cauliflower) — high in sulforaphane and fiber
- Colorful fruits (berries, citrus, pomegranate) — rich in antioxidants and vitamin C
- Whole grains (brown rice, quinoa, oats) — support microbiome diversity and bowel regularity
- Legumes (lentils, chickpeas, black beans) — top sources of folate and plant protein
- Oily fish (salmon, mackerel, sardines) — for omega-3s, unless following methionine restriction
Together, these foods reduce carcinogen exposure in the colon, support microbiome health, and supply key micronutrients. What you cut from your diet matters just as much.
Foods to Limit or Avoid
- Processed and red meats: The World Cancer Research Fund links processed meat to a 16% higher risk per 50g/day, and red meat to 12% per 100g/day. High-heat cooking generates carcinogenic heterocyclic amines and polycyclic aromatic hydrocarbons.
- Alcohol: Risk rises significantly above 30g/day. Gut bacteria convert ethanol to acetaldehyde, which depletes colonic folate and disrupts DNA methylation (abnormal gene expression patterns).
- Refined and processed foods: High sugar and saturated fat drive chronic inflammation, creating a metabolic environment that favors tumor growth.

Methionine Restriction
Cancer cells are highly dependent on the amino acid methionine for proliferation. Methionine restriction — achieved by reducing animal protein sources rich in methionine — is an emerging nutritional strategy in integrative oncology. Reducing protein intake in this way, especially animal protein, without causing malnutrition can support metabolic wellness, reduce IGF-1 levels, and is being explored alongside conventional care in ongoing research.
The "Hoffman effect" describes cancer cells' metabolic dependence on exogenous methionine due to defects in the methionine salvage pathway. At the cellular level, methionine restriction:
- Induces S/G2 phase cell cycle arrest, halting cancer cell division
- Depletes glutathione — the key defense cancer cells rely on to maintain elevated oxidative conditions, an area being actively investigated in nutritional oncology research
- Sensitizes tumors to chemotherapy and radiotherapy
NORI has developed protocols based on cycled methionine restriction combined with targeted nutraceuticals. This approach exploits cancer cells' amino acid vulnerability while minimizing impact on healthy tissue.
How to Choose Supplements Safely as a Colon Cancer Patient
The "More Is Better" Myth
High-dose antioxidant supplements (vitamins A, C, E, CoQ10, selenium) during active chemotherapy or radiation may interfere with treatment efficacy. Many cancer therapies depend on oxidative stress to kill cancer cells—antioxidants can protect cancer cells from this intended damage.
The American Cancer Society and NCI advise caution regarding high-dose antioxidants during treatment, noting they may lower the chance of staying cancer-free and increase mortality risk.
Critical rule: Disclose all supplements to your oncologist before, during, and after treatment.
Quality Factors to Look For
Third-party testing: Choose supplements verified by U.S. Pharmacopeia (USP), NSF International, or ConsumerLab.com. The FDA does not evaluate dietary supplements for efficacy before marketing.
Bioavailability-enhanced formulations: Especially important for curcumin and Vitamin D, where absorption is often the limiting factor.
Avoid proprietary blends: Undisclosed dosages make it impossible to assess safety and efficacy.
Integrative Oncology Programs
Integrative oncology programs combine research-backed nutritional protocols with conventional care, accounting for treatment stage, cancer type, and individual nutrient status.
NORI (Nutritional Oncology Research Institute) takes this further by formulating its own nutraceutical combinations in-house, grounded in over 20 years of research on methionine restriction and selective cancer cell targeting through oxidative stress mechanisms.
Test Before Supplementing
Knowing your baseline nutrient status is the starting point for any safe supplementation plan. Before adding supplements, get bloodwork to identify actual deficiencies:
- Vitamin D (serum 25(OH)D assay — recommended by the Endocrine Society before high-dose supplementation)
- Folate (especially relevant for colon cancer patients with dietary gaps)
- Magnesium (commonly depleted during chemotherapy)

Supplementing confirmed deficiencies is well-supported by evidence. Supplementing nutrients already at adequate levels offers less clear benefit and carries potential risk.
Conclusion
No supplement is a cure for colon cancer. However, the evidence supports targeted nutritional intervention—particularly Vitamin D, calcium, folate from food sources, bioavailability-enhanced curcumin, omega-3s, and synbiotics—as meaningful tools to support gut health, reduce inflammation, and improve quality of life when used correctly and under supervision.
Nutrition works best when it's built into a colon cancer care plan from the start, alongside medical treatment rather than layered on after the fact. For patients seeking a structured, research-backed approach, NORI's home-based nutritional support program offers personalized guidance on integrating diet and nutraceuticals into comprehensive care.
Schedule a free initial consultation by calling 800-634-3804 or emailing info@nutritionaloncology.net.
Frequently Asked Questions
What vitamin is deficient in colon cancer?
Vitamin D deficiency is the most common nutritional gap in colon cancer patients and is linked to poorer survival outcomes. Folate (B9) and magnesium deficiencies are also frequently observed and tied to colorectal cancer risk and progression.
What is the best diet to fight colon cancer?
A plant-based, high-fiber diet — vegetables, whole grains, legumes, and oily fish — has the strongest evidence base. Limit red and processed meats, alcohol, and refined sugars. Mediterranean and DASH dietary patterns are both associated with reduced colorectal cancer risk.
What foods should you avoid if you have colon cancer?
Avoid red and processed meats, alcohol, high-sugar processed foods, and excessive saturated fats. High-temperature cooking of meat generates known carcinogens, and alcohol disrupts folate metabolism and DNA methylation — both relevant to colorectal cancer risk.
Can supplements interfere with colon cancer treatment?
Yes. High-dose antioxidant supplements may reduce the effectiveness of chemotherapy and radiation, which depend partly on oxidative damage to kill cancer cells. Always inform your oncology team about any supplements you are taking — timing and dosage can affect outcomes.
Is curcumin effective for colon cancer patients?
Early lab and clinical evidence supports curcumin's anti-inflammatory and wellness-supporting effects in colorectal cancer, but standard curcumin powder is poorly absorbed. Bioavailability-enhanced formulations — such as those combining piperine — and medical supervision are necessary for any meaningful benefit.
What is the new treatment for colon cancer?
Conventional advances include targeted therapies (BRAF and KRAS G12C inhibitors) and immunotherapy for MSI-H/dMMR tumors. On the integrative side, methionine-restricting diets and targeted nutraceutical protocols — such as those developed through NORI's research — are an emerging area of supportive care that targets metabolic vulnerabilities in cancer cells.


