
Introduction
Iron deficiency affects approximately 58% of cancer patients at diagnosis, with functional iron deficiency—where inflammation locks iron away from circulation—being the dominant form in active cancer. The prevalence climbs even higher during chemotherapy, with up to 67% of patients developing anemia over a six-month treatment period.
Choosing the right iron supplement as a cancer patient is far from straightforward. The same iron that combats crushing fatigue can also promote tumor growth under certain conditions.
Research has found associations between iron supplementation during breast cancer chemotherapy and adverse outcomes, which is why physician-guided supplementation is essential — not something to navigate alone.
This guide walks through why iron deficiency develops in cancer, the key differences between oral and intravenous iron, which supplement forms absorb best, and when supplementation should be avoided entirely.
TL;DR
- Functional iron deficiency affects most cancer patients due to inflammation-driven iron blockade (hepcidin), causing fatigue and reduced treatment tolerance
- Oral iron absorption is severely impaired (more than 95% excreted) during active cancer, making IV iron the clinically preferred approach for most patients
- Gentler oral forms like bisglycinate or liposomal iron suit limited situations — remission, mild deficiency, or low systemic inflammation
- Skip iron supplementation during active infection, with ferritin above 800 ng/mL, or with blood cancers — always under specialist oversight
- Confirm iron status with bloodwork (ferritin, TSAT, CBC) before supplementing and coordinate directly with your oncology team
Why Iron Deficiency Is So Common in Cancer Patients
Cancer patients face two distinct types of iron deficiency. Absolute iron deficiency means your body's iron stores are truly depleted — ferritin below 30 ng/mL signals empty reserves.
Functional iron deficiency is far more common in cancer: ferritin appears normal or even high, but inflammation traps that iron in storage cells, making it unavailable for red blood cell production.
The Hepcidin Blockade
Inflammatory cytokines like IL-6 trigger hepcidin production in your liver. Hepcidin then degrades ferroportin—the protein that moves iron out of storage cells and into circulation. When ferroportin breaks down, iron remains trapped in macrophages and intestinal cells, unable to reach the bone marrow where red blood cells are made.
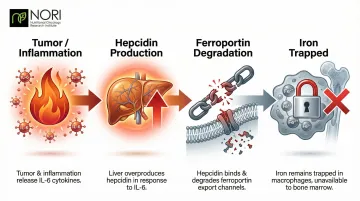
Over 50% of cancer patients show transferrin saturation (TSAT) below 20% — the hallmark of functional iron deficiency.
Additional causes of iron loss:
- Chronic bleeding from GI or gynecological tumors
- Chemotherapy-induced bone marrow suppression
- Surgical blood loss
- Reduced dietary intake from nausea, appetite loss, or mucositis
Why Standard Ferritin Testing Misleads
Ferritin is an acute-phase protein — it rises with inflammation regardless of actual iron stores. A cancer patient with ferritin of 400 ng/mL may still be severely iron deficient if inflammation is driving that number up. TSAT below 20% is the more reliable marker in oncology populations.
Consequences of untreated iron deficiency:
- Profound fatigue and cognitive impairment
- Reduced physical fitness and exercise capacity
- Impaired chemotherapy adherence
- Increased risk of blood transfusions
- Diminished quality of life and treatment efficacy
Oral vs. Intravenous Iron: What Cancer Patients Need to Know
Why Oral Iron Often Fails
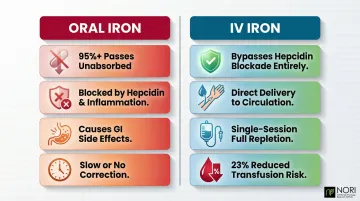
The same hepcidin mechanism that traps stored iron also blocks intestinal absorption. More than 95% of oral iron passes through unabsorbed in patients with active cancer-related inflammation. You're essentially excreting expensive supplements with minimal benefit.
When oral iron is appropriate:
- Complete remission with no active inflammation (CRP below 5 mg/L)
- Mild absolute iron deficiency (ferritin below 30 ng/mL)
- No urgent need to correct anemia quickly
- Patients who cannot access IV iron therapy
Why IV Iron Is Clinically Preferred
Intravenous iron bypasses the absorptive blockade entirely by delivering iron directly to macrophages and circulation. Modern formulations like ferric carboxymaltose (FCM) and ferric derisomaltose (FDI) allow single high-dose infusions (up to 1,000 mg in 15–20 minutes) with favorable safety profiles.
Key clinical advantages include:
- Circumvents hepcidin-driven absorption blockade entirely
- Delivers iron directly into circulation for faster utilization
- Reduces transfusion risk by 23% when combined with erythropoiesis-stimulating agents (ESAs)
- Enables full repletion in a single infusion session
The Risk-Benefit Context
The clinical benefits of IV iron don't eliminate the need for careful risk assessment. Observational research from the DELCaP study found associations between iron supplementation during breast cancer chemotherapy and a 79% increased risk of disease recurrence (HR 1.79). While this doesn't prove causation, it reinforces a critical principle: iron therapy in cancer requires physician guidance aligned with your treatment plan—never self-supplementation based on fatigue alone.
Best Iron Supplement Forms for Cancer Patients
The "best" oral iron balances bioavailability with GI tolerability, doesn't interfere with ongoing treatment, and is used only when clinically indicated. Here are the five main forms.
Iron Bisglycinate (Chelated Iron)
Iron bisglycinate binds iron to two glycine molecules, allowing absorption through a separate pathway that bypasses mineral competition and reduces GI irritation compared to iron salts.
For cancer patients already managing nausea, mucositis, or chemotherapy side effects, this gentler GI profile matters. Research shows higher bioavailability per milligram than ferrous sulfate, meaning lower doses achieve similar results.
| Aspect | Details |
|---|---|
| Form | Chelated (bisglycinate) |
| Elemental Iron | 14-28 mg per capsule |
| GI Tolerability | High—significantly less constipation and nausea |
| Best For | Chemotherapy patients with active GI side effects requiring mild oral supplementation |
Ferrous Gluconate
This iron salt contains lower elemental iron per tablet than ferrous sulfate, making it a gentler option used in clinical oncology settings for mild absolute iron deficiency.
Lower elemental iron content means fewer GI side effects while still providing meaningful supplementation — making it a practical starting point for cancer patients who can tolerate oral iron.
| Aspect | Details |
|---|---|
| Form | Ferrous salt |
| Elemental Iron | ~38 mg per 324 mg tablet |
| GI Tolerability | Moderate—better than ferrous sulfate, more side effects than chelated forms |
| Best For | Patients with mild iron deficiency in remission or low-grade inflammation |
Ferrous Sulfate
The most widely studied and prescribed oral iron supplement. Ferrous sulfate has the highest elemental iron concentration per dose and serves as the reference standard in oncology anemia guidelines.
Despite its GI side effects, it remains the clinical reference point in research and the most affordable widely available option — a real consideration for long-term management.
| Aspect | Details |
|---|---|
| Form | Ferrous salt |
| Elemental Iron | ~65 mg per 325 mg tablet |
| GI Tolerability | Low to moderate—commonly causes constipation, nausea, dark stools |
| Best For | Patients in remission with confirmed absolute iron deficiency, low inflammation, and ability to tolerate GI effects |
Ferrous sulfate increases GI side effects significantly versus placebo (OR 2.32) and versus IV iron (OR 3.05).
Liposomal Iron
Liposomal iron encapsulates ferric pyrophosphate within a phospholipid vesicle, protecting the GI mucosa from direct iron contact and allowing absorption in the small intestine.
It achieves comparable or superior absorption to ferrous sulfate at lower doses, with far fewer GI side effects. Reduced free-iron release may also lower oxidative stress — a consideration worth noting for cancer patients.
| Aspect | Details |
|---|---|
| Form | Liposomal encapsulation |
| Elemental Iron | 14-30 mg per sachet or capsule |
| GI Tolerability | Very high—minimal direct GI contact |
| Best For | Cancer patients with confirmed need for oral supplementation who cannot tolerate standard iron forms |

When oral iron isn't enough — or when hepcidin-driven inflammation blocks absorption entirely — IV iron becomes the appropriate clinical intervention.
Intravenous Iron (Clinical Setting)
IV formulations — ferric carboxymaltose (FCM), iron sucrose, ferric derisomaltose (FDI) — are the gold standard for iron repletion in active cancer patients. Clinicians administer these in controlled settings; they're not available over the counter.
IV iron completely bypasses the hepcidin blockade. Ferric carboxymaltose delivers up to 1000 mg in a single 15-minute infusion, supporting faster hemoglobin recovery and reduced transfusion requirements. Multiple RCTs support its use both alone and alongside ESAs.
Important safety note: FCM carries a 75% risk of hypophosphatemia compared to 7.9% for FDI, making formulation choice clinically significant.
| Aspect | Details |
|---|---|
| Form | Ferric hydroxide complexes |
| Elemental Iron | 50-100 mg/mL; doses of 200-1000 mg per infusion |
| GI Tolerability | N/A—bypasses GI tract; rare infusion reactions; contraindicated during active infection |
| Best For | Active cancer patients with functional or absolute iron deficiency, chemotherapy-induced anemia, pre-surgical optimization |
How to Choose the Right Iron Supplement (and When to Avoid It)
Key Evaluation Factors
Before choosing any iron supplement, confirm:
- Type of deficiency: Absolute (ferritin below 30 ng/mL) vs. functional (TSAT below 20%, ferritin above 100 ng/mL)
- Inflammation status: Check C-reactive protein (CRP)—high CRP predicts oral iron failure
- ESA therapy: If you're receiving erythropoiesis-stimulating agents, IV iron dramatically enhances response
- Treatment phase: Active chemotherapy vs. remission vs. pre-surgical preparation
When to Avoid Iron Supplementation
Absolute contraindications:
- Active infections—iron feeds pathogens
- Ferritin above 800 ng/mL (withhold) or above 1000 ng/mL (contraindicated per clinical guidelines)
- Blood cancers such as leukemia—supplementation can increase infection risk without strict physician oversight
Because iron decisions depend heavily on lab values, cancer type, and treatment phase, working with a knowledgeable practitioner matters. NORI's nutritional oncology program provides personalized supplement and dietary guidance tailored to your specific situation—including a free initial consultation. Call 800-634-3804 to get started.
Dietary Ways to Boost Iron During Cancer Treatment
Heme vs. Non-Heme Iron
Heme iron from animal sources (red meat, poultry, fish) absorbs at 15–35%. Non-heme iron from plant sources (lentils, spinach, white beans, tofu, fortified cereals) absorbs at just 2–20%—an important distinction for patients avoiding animal products.
Proven Absorption Strategies
To increase non-heme iron absorption:
- Pair iron-rich plant foods with vitamin C sources (citrus, bell peppers, strawberries, tomatoes)
- Increasing vitamin C from 25 mg to 1000 mg raised iron absorption from 0.8% to 7.1%
- Practical pairings: spinach salad with lemon juice, tofu stir-fry with broccoli and tomatoes
Common absorption inhibitors to avoid:
- Tannins in coffee and tea — 50 mg of polyphenols reduced bioavailability by 18%
- Dairy calcium reduces absorption by 18–27%
- Phytates in unsoaked legumes and cereal bran (dose-dependent inhibition)

Reality Check for Cancer Patients
These strategies work best as a foundation. For patients with active inflammation, dietary iron alone is unlikely to correct iron deficiency anemia — but optimizing food sources can meaningfully reduce deficiency severity and is a practical starting point when oral supplements cause intolerance or fail to absorb.
Frequently Asked Questions
Is it safe for cancer patients to take iron supplements?
Safety depends on your iron deficiency type, inflammation status, ferritin level, and treatment phase. Oral supplements are generally safer in remission while IV iron is preferred during active cancer. Self-supplementing without confirmed lab-based deficiency is not recommended.
What is the relationship between iron and cancer?
Iron deficiency causes anemia that worsens treatment outcomes, but excess free iron can fuel tumor cell growth and support pathogens. This dual risk is why supplementation requires careful clinical assessment — not routine use.
Do people with cancer have low iron?
Yes, iron deficiency is very common. Prevalence varies by cancer type: 63% in pancreatic cancer, 52% in colorectal cancer, and 51% in lung cancer. However, "low iron" may not show up on standard ferritin tests due to inflammation falsely elevating ferritin levels.
How to increase iron while on chemo?
IV iron is the clinically preferred method during active chemotherapy, since oral absorption is severely impaired by inflammation. Dietary strategies — heme iron sources and vitamin C pairing — offer supplemental support. Always confirm iron status with TSAT before starting any intervention.
Who should avoid iron supplements?
Patients with active infections, very high ferritin (above 800–1000 ng/mL), iron overload conditions, or certain blood cancers like leukemia should avoid supplementation without physician oversight. Those on specific chemotherapy regimens may also need to pause supplementation.
What supplements should cancer patients avoid?
Be cautious with high-dose antioxidants (vitamins A, C, E, CoQ10, beta-carotene) during chemotherapy as they may reduce drug efficacy. Iron supplements without confirmed deficiency also carry risk. Always disclose all supplements to your oncology team before use.


