
Introduction
Immunotherapy has redefined what's possible in cancer care, delivering meaningful survival gains with less toxicity than chemotherapy. In advanced non-small cell lung cancer, pembrolizumab delivered a median progression-free survival of 10.3 months versus 6.0 months for chemotherapy, while 5-year overall survival rates for nivolumab reached 13.4% compared to just 2.6% for docetaxel.
Despite these advances, many patients still struggle with suboptimal responses. What's increasingly clear is that nutrition plays a direct role in how well immunotherapy works.
The National Cancer Institute now acknowledges that diet influences immunotherapy efficacy, stating that "consuming a diet rich in fiber, like fruits, vegetables, and legumes, could improve your ability to respond to immunotherapy" by shaping the gut microbiome.
This article covers the specific dietary patterns, nutrients, and strategies that can enhance immunotherapy response, reduce treatment side effects, and support long-term survival—and what to avoid to prevent undermining your treatment.
TLDR
- Dietary fiber (≥20g/day) improves progression-free survival in immunotherapy patients by up to 30%
- Mediterranean diet linked to 77% objective response rate in melanoma patients on checkpoint inhibitors
- Normal Vitamin D levels increase response rates from 36% to 56% during anti-PD-1 therapy
- High-dose antioxidant supplements may interfere with immunotherapy by neutralizing cancer-killing reactive oxygen species
- Methionine restriction synergizes with checkpoint inhibitors as an advanced nutritional strategy
Why Immunotherapy Patients Have Unique Nutritional Needs
Unlike chemotherapy—which kills rapidly dividing cells indiscriminately—immunotherapy activates your immune system to recognize and destroy cancer cells. This fundamental difference creates specific nutritional needs centered on supporting immune cell activity, reducing inflammatory burden, and maintaining a healthy gut microbiome.
Immunotherapy Side Effects Differ from Chemotherapy
Immune-related adverse events (irAEs) create distinct nutritional challenges. Immune-related enterocolitis typically develops 5 to 10 weeks after checkpoint inhibitor treatment begins, though it can appear months later. Incidence of diarrhea or colitis ranges from 10% with anti-PD-1 therapy to 35% with anti-CTLA-4 therapy, with combination therapy reaching 32%.
These rates aren't just statistics: colitis-driven diarrhea, fatigue that suppresses appetite, and persistent nausea can each erode nutrient intake quickly if not addressed before they escalate.
Malnutrition Undermines Treatment Success
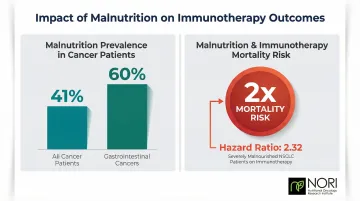
Using the Global Leadership Initiative on Malnutrition (GLIM) criteria, 41% of cancer patients experience malnutrition, with rates reaching 60% in gastrointestinal cancers. This matters because malnutrition during cancer treatment is associated with worse treatment tolerance, more dose reductions, and significantly lower survival rates.
In NSCLC patients treated with immunotherapy, severe malnutrition at baseline was associated with worse treatment efficacy and lower survival (hazard ratio 2.32). A hazard ratio of 2.32 means severely malnourished patients faced more than double the mortality risk — which is why nutritional status should be assessed and addressed at the start of treatment, not as an afterthought.
How Your Diet Shapes Immune Response and the Tumor Microenvironment
The Gut-Immune Axis: Your Body's Largest Immune Organ
The gastrointestinal tract houses up to 70% of your body's lymphocyte population and approximately 70% of plasma antibody-producing cells. This massive immune presence means your gut microbiome directly influences the activity of T cells—including the CD8+ cytotoxic T cells that immunotherapy depends on to destroy cancer.
Short-chain fatty acids (SCFAs) produced when gut bacteria ferment dietary fiber enhance CD8+ T cell memory and sustain wellness-supporting immunity by fueling oxidative phosphorylation. When you consume adequate fiber, beneficial bacteria produce butyrate and other SCFAs that literally enhance your immune cells' health-supporting capacity.
Mediterranean Diet: 77% Response Rate in Melanoma Patients
In a multicenter study of 91 advanced melanoma patients in the UK and Netherlands, high adherence to a Mediterranean diet was associated with a 77% probability of objective response and a 74% probability of 12-month progression-free survival. This wasn't a marginal benefit—it was a substantial improvement in treatment outcomes achieved through dietary choices alone.
The Mediterranean pattern emphasizes:
- Vegetables, fruits, whole grains, and legumes
- Fish rich in omega-3 fatty acids, olive oil, and nuts
- Foods that reduce systemic inflammation
- Diverse fiber sources that feed SCFA-producing gut bacteria
Dietary Fiber: 30% Lower Risk of Progression Per 5g Daily Increase
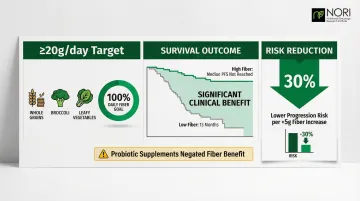
In a cohort of 128 melanoma patients on checkpoint inhibitors, sufficient fiber intake (≥20g/day) was associated with improved progression-free survival. Median PFS wasn't even reached in the sufficient fiber group versus 13 months for insufficient intake. Every 5g/day increase in fiber corresponded to a 30% lower risk of progression or death.
The greatest benefit appeared in patients with sufficient fiber intake who did not use over-the-counter probiotic supplements. Concurrent probiotic use actually negated the fiber benefit and was associated with lower response rates. What you eat on the other side of that equation matters just as much.
Pro-Inflammatory Diets Create Immunosuppressive Conditions
High consumption of processed foods, refined sugars, and saturated fats drives systemic inflammation that promotes an immunosuppressive tumor microenvironment. Preclinical research shows that high-fat, obesity-inducing diets upregulate leptin, which drives accumulation of immunosuppressive Myeloid-Derived Suppressor Cells (MDSCs) and promotes CD8+ T cell exhaustion.
These cellular conditions directly impair checkpoint inhibitor function — making it harder for your immune system to recognize and attack cancer cells regardless of treatment dose.
Polyphenols and PD-L1 Expression: An Emerging Frontier
Specific polyphenols found in plant foods—including curcumin from turmeric, EGCG from green tea, resveratrol from grapes, and apigenin from parsley—have shown preliminary evidence of modulating PD-L1 expression in cancer cells. Curcumin and apigenin inhibit IFN-γ-induced STAT1 activation, thereby downregulating PD-L1 expression, while resveratrol targets PD-L1 by disrupting its glycosylation and dimerization.
The evidence is still largely preclinical, but polyphenol-rich whole foods appear to act on some of the same molecular targets as PD-1/PD-L1 checkpoint inhibitors — a convergence worth tracking as clinical data matures.
Dietary Patterns That Enhance Immunotherapy Response
Mediterranean Diet: The Most Evidence-Backed Pattern
The Mediterranean dietary pattern has the strongest clinical evidence for improving immunotherapy outcomes. This approach emphasizes:
- Vegetables and fruits: multiple servings daily for fiber, polyphenols, and micronutrients
- Whole grains: brown rice, quinoa, whole wheat, oats
- Legumes: lentils, chickpeas, beans — excellent fiber sources
- Fish: salmon, sardines, mackerel, rich in omega-3 fatty acids
- Olive oil: primary fat source with anti-inflammatory compounds
- Nuts and seeds: moderate portions for healthy fats and minerals
This pattern consistently produces low Dietary Inflammatory Index scores, reduces systemic inflammation, and supports the diverse gut microbiome that correlates with better checkpoint inhibitor response.
High-Fiber Eating: Foundation for Gut Health
Aim for at least 20g of dietary fiber daily to meaningfully impact immunotherapy outcomes. Practical high-fiber choices include:
- Legumes and lentils: black beans, kidney beans, chickpeas (15g per cup)
- Cruciferous vegetables: broccoli, Brussels sprouts, cauliflower
- Whole grains: oats, barley, brown rice, quinoa
- Berries: raspberries, blackberries, strawberries
- Vegetables: artichokes, sweet potatoes, carrots
High dietary fiber intake promotes abundance of SCFA-producing bacteria like Ruminococcaceae, which correlates with increased CD4+ and CD8+ T cell infiltration in the tumor microenvironment during anti-PD-1 therapy.
Methionine Restriction: Advanced Nutritional Intervention
Most dietary strategies work around cancer — methionine restriction works against it. In colorectal cancer mouse models, dietary methionine restriction increased tumor MHC-I and PD-L1 expression. When combined with anti-PD-1 and anti-CTLA-4 checkpoint inhibitors, it was five times more effective at reducing tumor size than checkpoint inhibitors alone.
Restricting methionine by approximately 80% triggers three distinct effects:
- Enhances antigen presentation on cancer cells, making them more visible to the immune system
- Reprograms tumor-associated macrophages toward wellness-supporting behavior
- Enhances the effect of immune checkpoint blockade rather than working in parallel to it

NORI's protocol centers on cycled methionine restriction combined with targeted nutraceuticals, developed over 20 years of focused research. Their home-based support program includes individualized guidance on implementing this approach safely — call 800-634-3804 for a free consultation.
Ketogenic Diet: Preclinical Promise Requiring Medical Supervision
In mouse models, a ketogenic diet or oral supplementation with the ketone body 3-hydroxybutyrate prevented checkpoint inhibitor-linked upregulation of PD-L1 on myeloid cells, expanded CXCR3+ T cells, and enhanced the effect of anti-PD-1 therapy to slow aggressive tumor growth.
The proposed mechanism centers on reducing glucose availability, which lowers lactate production by cancer cells. Lactate inhibits NK cell and dendritic cell function, so reducing it may enhance immune surveillance. However, evidence remains largely preclinical, and this diet requires medical supervision to prevent malnutrition and ensure adequate protein intake.
Maintaining Adequate Protein: A Critical Balance
While methionine restriction shows promise, general protein adequacy remains essential to prevent cachexia and maintain immune cell production. ESPEN guidelines recommend 1.0–1.5 g/kg/day of protein for adult cancer patients to support muscle protein synthesis.
The distinction between protein quality matters in therapeutic dietary strategies: methionine-rich animal proteins (beef, eggs, dairy) versus plant-based proteins (legumes, quinoa, nuts) offer different methionine loads, allowing for adequate total protein while managing methionine intake.
Key Nutrients and Natural Compounds That Potentiate Immunotherapy
Vitamin D: 56% Response Rate vs. 36% in Deficient Patients
According to a study of 200 advanced melanoma patients on anti-PD-1 therapy, those with normal serum Vitamin D levels achieved an objective response rate of 56.0% compared to 36.2% in deficient patients. Median progression-free survival was significantly longer in the normal Vitamin D group: 11.25 months versus 5.75 months.
Vitamin D deficiency is common in cancer patients, and maintaining adequate levels—either through baseline sufficiency or supplementation—is associated with improved immunotherapy prognosis. Work with your oncologist to check levels and supplement appropriately.
Omega-3 Fatty Acids: Modulating Inflammation and Macrophage Polarization
Beyond fat-soluble vitamins, omega-3 fatty acids offer a distinct immune-modulating mechanism. EPA and DHA modulate pro- and anti-inflammatory cytokines and shift macrophage polarization toward wellness-supporting phenotypes. Direct checkpoint inhibitor data is still emerging, but a 2011 study in advanced NSCLC patients found that 2.5g/day EPA+DHA raised response rates to platinum-based chemotherapy from 25.8% to 60.0%.
Good dietary sources include:
- Salmon, sardines, and mackerel
- Algae-based supplements (suitable for plant-based diets)
Polyphenols from Whole Foods: Suppress Immune-Suppressive Signaling
Curcumin (turmeric), EGCG (green tea), resveratrol (red grapes and berries), and anthocyanins (berries) suppress key immune-suppressive signaling pathways—STAT3, NF-κB, JAK2/STAT1—that drive PD-L1 expression on tumor cells.
These compounds are best obtained from whole foods. High-dose supplements may not mirror these effects and carry interaction risks. Practical ways to incorporate them daily:
- Add turmeric to cooking and soups
- Drink green tea regularly (2–3 cups per day)
- Include berries and red grapes as daily snacks
Short-Chain Fatty Acids: Butyrate and Progression-Free Survival
Fermentable fiber is converted by gut bacteria into butyrate, which modulates immune responses in ways that improve checkpoint inhibitor outcomes. In a cohort of 52 solid tumor patients treated with PD-1 inhibitors, high fecal concentrations of butyric acid were significantly associated with longer progression-free survival (hazard ratio 0.31).
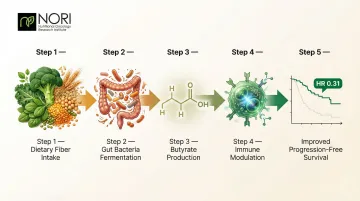
Butyrate cannot be taken as a direct supplement. Instead, feed the bacteria that produce it through consistent fiber intake from vegetables, legumes, and whole grains.
What to Avoid: Foods and Supplements That May Undermine Immunotherapy
High-Dose Antioxidant Supplements: The Paradox
While antioxidant-rich whole foods support immune function, high-dose antioxidant supplements — Vitamin E, beta-carotene, N-acetylcysteine — may interfere with immunotherapy.
While antioxidant-rich whole foods support immune function, high-dose antioxidant supplements — Vitamin E, beta-carotene, N-acetylcysteine — may interfere with immunotherapy. Randomized clinical trials have demonstrated that high-dose antioxidant supplements may increase cancer incidence and progression, potentially by neutralizing the reactive oxygen species that activated T cells use to kill cancer cells.
Activated immune cells generate reactive oxygen species as part of their cancer-killing mechanism. In high doses, antioxidant supplements may shield cancer cells from that attack — the opposite of what you need.
Common high-dose supplements to discuss with your oncologist before continuing:
- Vitamin E (above dietary levels)
- Beta-carotene supplements
- N-acetylcysteine (NAC)
- High-dose Vitamin C (IV or supplemental)
High Saturated Fat and Ultra-Processed Food Diets
High-fat diet-induced obesity impairs CD8+ T cell function and infiltration in the tumor microenvironment, accelerating tumor growth in animal models. A diet high in processed foods, refined sugars, and saturated fats increases systemic inflammation and can blunt immunotherapy response.
A structured ketogenic diet may offer specific benefits through different metabolic mechanisms. An unstructured Western diet heavy in processed foods does not — it drives chronic inflammation without the metabolic discipline that gives therapeutic ketosis its potential value.
Antibiotics: Devastating Impact on Gut Microbiota
Broad-spectrum antibiotic use significantly reduces immunotherapy efficacy by depleting gut microbiota diversity. Mouse studies showed that CTLA-4 blockade failed completely in antibiotic-treated mice, though efficacy was restored by oral administration of beneficial bacteria like Bacteroides fragilis.
If you need antibiotics during immunotherapy, discuss this carefully with your care team and consider gut microbiome recovery strategies afterward, including high-fiber foods and targeted probiotics under medical guidance.
Practical Nutrition Tips for Managing Immunotherapy Side Effects
Immune-Related Colitis and Diarrhea
For Grade 1 immunotherapy-induced diarrhea or colitis, clinical guidelines recommend dietary modifications including a low-residue, bland, or lactose-free diet. The BRAT approach provides relief:
- Bananas — easy to digest and restore potassium
- Rice — white rice during acute flares; switch to brown once recovered
- Applesauce — gentle on the gut with useful nutrients
- Toast — plain white bread only while symptomatic

Ensure adequate electrolyte replacement through broths, coconut water, or electrolyte drinks. Contact your care team if diarrhea persists beyond 48 hours or worsens.
Probiotic supplements require caution during immunotherapy — they may interfere with the microbiome responses that dietary fiber helps support. Always check with your oncologist before adding them.
Fatigue and Appetite Loss
Combat treatment-related fatigue and poor appetite with these strategies:
- Eat every 2–3 hours in small portions rather than forcing three full meals
- Choose calorie-dense foods: nut butters, avocado, eggs, and Greek yogurt deliver nutrition without large volume
- Blend a protein smoothie with Greek yogurt, fruit, spinach, and nut butter when solid food feels difficult
- Move a little daily — ASCO recommends aerobic or resistance exercise tailored to your abilities to reduce fatigue, and even short walks can restore appetite
Working with a Nutritional Oncology Specialist
Self-managing side effects through diet is a strong foundation, but personalized guidance makes a measurable difference. NORI (Nutritional Oncology Research Institute) offers a home-based support program that pairs individualized dietary protocols with nutraceuticals developed through focused cancer nutrition research. Free initial consultations are available by phone at 800-634-3804 or by email at info@nutritionaloncology.net.
Frequently Asked Questions
What is the best diet for cancer patients on immunotherapy?
The Mediterranean diet—rich in vegetables, fruits, whole grains, fish, and legumes—has the strongest evidence for improving immunotherapy response, with 77% objective response rates in melanoma patients. Complement this with adequate fiber intake (≥20g/day) to support a healthy gut microbiome.
Does diet actually affect how well immunotherapy works?
Yes. Multiple clinical studies have now demonstrated that diet quality and gut microbiome composition directly influence immunotherapy response rates and progression-free survival. In melanoma patients, sufficient fiber intake (≥20g/day) was associated with a 30% lower risk of progression per 5g increase.
What foods should cancer patients avoid during immunotherapy?
Limit or avoid:
- High-dose antioxidant supplements (Vitamin E, beta-carotene)
- Ultra-processed foods and high-saturated-fat foods
- Alcohol and unnecessary antibiotic use
These deplete beneficial gut bacteria or shield cancer cells from treatment.
How can a cancer patient increase their lifespan during and after treatment?
Follow an anti-inflammatory diet like the Mediterranean diet, stay physically active, and support gut health through fiber-rich foods. Working closely with an integrative oncology team ties these strategies together and has shown measurable improvements in survival outcomes.
Can cancer survivors live a normal life after treatment?
Many cancer survivors live full, active lives — particularly those who maintain healthy nutrition, regular exercise, and follow-up monitoring. The choices you make during and after treatment have a direct impact on long-term quality of life and recurrence risk.


