The treatment landscape has expanded dramatically over the past decade. Immunotherapy, targeted therapy, radiation, surgery, and nutritional support have each evolved from experimental approaches into proven, evidence-based options that give patients and oncologists far more to work with than a generation ago.
This guide covers what's available beyond chemotherapy—particularly for patients who want to understand all their options, those concerned about chemotherapy's side effects, and those looking to complement a conventional plan with integrative support. Whether you're newly diagnosed or weighing next steps after initial treatment, these alternatives deserve your careful consideration.
TLDR
- Immunotherapy is now a standard first-line option for many NSCLC patients with high PD-L1 expression, often without any chemotherapy
- Targeted therapies match drugs to specific genetic mutations (EGFR, ALK, KRAS) and typically come in pill form with fewer side effects
- Radiation (especially SBRT) and surgery can be curative for early-stage lung cancer without chemotherapy
- Methionine restriction and nutraceutical protocols target metabolic vulnerabilities in cancer cells — an evidence-based nutritional strategy, not just a supplement afterthought
- Skipping biomarker testing before treatment means potentially missing a targeted therapy or immunotherapy your tumor biology is uniquely suited for
Immunotherapy for Lung Cancer: Using Your Immune System as a Weapon
How Checkpoint Inhibitors Work
Cancer cells use proteins like PD-L1 to "hide" from your immune system. These proteins block T-cells from recognizing and attacking tumors. Immune checkpoint inhibitors like pembrolizumab (Keytruda) remove this block, allowing your immune system to identify and destroy cancer cells that were previously invisible to it.
Unlike chemotherapy — which poisons all rapidly dividing cells — immunotherapy trains your own immune system to recognize and eliminate the tumor directly.
Who Qualifies for Chemotherapy-Free Immunotherapy
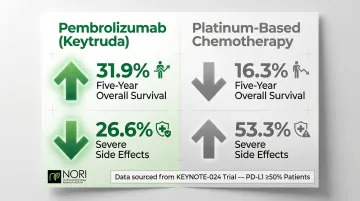
PD-L1 testing determines your eligibility. Patients with a PD-L1 tumor proportion score (TPS) of 50% or higher may qualify for pembrolizumab monotherapy as first-line treatment—no chemotherapy involved.
The KEYNOTE-024 trial demonstrated that first-line pembrolizumab nearly doubled the 5-year overall survival rate (31.9% vs 16.3%) compared to platinum-based chemotherapy in patients with PD-L1 ≥50%. Severe side effects dropped sharply as well — 26.6% versus 53.3% with chemotherapy.
FDA-Approved Immunotherapy Options
Four checkpoint inhibitors are now approved for NSCLC:
| Agent | Primary Use | Approval Year |
|---|---|---|
| Pembrolizumab | First-line monotherapy (PD-L1 ≥1%); combination with chemotherapy; adjuvant after surgery | 2014/2023 |
| Nivolumab | First-line combination with ipilimumab (PD-L1 ≥1%); neoadjuvant with chemotherapy | 2014/2024 |
| Atezolizumab | First-line monotherapy (high PD-L1); combination with chemotherapy; adjuvant post-surgery | 2016/2021 |
| Durvalumab | Consolidation after chemoradiation for unresectable Stage III NSCLC | 2018/2024 |
Chemotherapy-Free Radiation Plus Immunotherapy
Emerging research shows promise for locally advanced (Stage III) NSCLC patients who cannot tolerate chemotherapy or prefer to avoid it. Four Phase II trials have tested radiation combined with immunotherapy alone:
- SPRINT trial: 76% one-year progression-free survival in patients with PD-L1 ≥50% receiving radiation plus pembrolizumab
- DOLPHIN trial: 72.1% one-year PFS with radiation plus durvalumab in patients with PD-L1 ≥1%
- SPIRAL-RT: 39.1% one-year PFS in patients ineligible for chemoradiation
- DUART trial: 47% one-year PFS in patients ineligible for chemotherapy
These biomarker-selected approaches show that for the right patient, chemotherapy may be unnecessary even in advanced disease.
Side Effect Comparison
Most immunotherapy patients avoid the signature toxicities of chemotherapy: no hair loss, severe nausea, or bone marrow suppression. Instead, the side effect profile shifts toward immune-related reactions — a different category of risk that requires different monitoring.
The most common immune-related adverse events include:
- Pneumonitis (lung inflammation)
- Colitis (intestinal inflammation)
- Endocrine dysfunction (thyroid, pituitary)
- Fatigue
Most are manageable and reversible with corticosteroids when caught early. Reporting new symptoms promptly gives your oncology team the best chance to intervene before they escalate.
Targeted Therapy: Precision Medicine Matched to Your Tumor's Genetics
How Targeted Therapy Differs
Targeted therapy requires biomarker testing of your tumor tissue to identify specific mutations or alterations driving cancer growth. The drug chosen matches that vulnerability rather than attacking all rapidly dividing cells.
Where chemotherapy hits every fast-dividing cell in the body, targeted therapy homes in on the specific molecular flaw keeping your cancer alive — leaving healthy tissue largely out of the crossfire.
Major Actionable Mutations and Corresponding Drugs
Comprehensive genomic profiling shows that up to 66% of lung adenocarcinomas harbor targetable alterations:
EGFR mutations (11-18% of adenocarcinomas)
- Osimertinib, erlotinib, gefitinib
- Particularly common in never-smokers and Asian populations
KRAS mutations (32-46% overall; G12C subtype 13-20%)
- Sotorasib, adagrasib specifically for G12C
- Recent breakthrough after decades without KRAS-targeted options
ALK rearrangements (1-1.3%)
- Alectinib, lorlatinib
- Highly responsive with dramatic tumor shrinkage
ROS1 fusions (0.3-1.7%)
- Crizotinib, entrectinib, repotrectinib
BRAF V600E (1.8-4.6%)
- Dabrafenib plus trametinib combination
RET fusions (0.7-0.9%)
- Selpercatinib, pralsetinib
MET exon 14 (2.9-4.5%)
- Capmatinib, tepotinib
The Patient Experience: Pills Instead of Infusions
Most targeted therapies come as daily oral pills rather than IV infusions. This makes an enormous practical difference. A 2019 study found that 77% of patients preferred oral therapy over IV, citing less disruption to daily life (70.4%) and avoiding infusion problems (56.3%).
Quality of life data supports this preference. In the PROFILE 1014 trial, first-line crizotinib showed significantly greater improvements in global quality of life and lung cancer symptoms compared to chemotherapy. The LUX-Lung 6 trial demonstrated similar benefits for afatinib versus cisplatin/gemcitabine.

The Resistance Challenge
That said, these practical benefits come with a significant caveat. Targeted therapies eventually face acquired resistance as cancer cells evolve, and most patients experience 1-3 years of response before progression.
Common resistance mechanisms:
- EGFR resistance: MET amplification (16%), EGFR C797S mutations (6%)
- ALK resistance: G1202R solvent-front mutation
- ROS1 resistance: G2032R mutation
- RET resistance: G810 solvent-front mutations
The good news: newer generations of inhibitors have been developed specifically to overcome these mechanisms. Osimertinib overcomes T790M resistance to first-generation EGFR inhibitors, while lorlatinib retains activity against G1202R ALK mutations. When resistance does occur, re-biopsy or liquid biopsy can reveal which new mechanism has emerged and guide the next treatment step.
Who Does NOT Qualify
You're unlikely to benefit from targeted therapy if you have:
- Squamous cell histology (lower mutation rates)
- Small cell lung cancer (different biology)
- No identified actionable mutations
This underscores why comprehensive molecular profiling before starting any treatment is critical. Testing that finds no current targets may lead to retesting later or consideration of clinical trials.
Surgery and Radiation Therapy: Chemotherapy-Free Options for Eligible Patients
Surgery for Early-Stage Disease
Lobectomy—removal of one lung lobe containing the tumor—remains the gold standard for Stage I-II NSCLC. For tumors over 2cm with solid appearance, NCCN and ESMO guidelines recommend surgical resection as primary treatment.
Minimally invasive techniques have significantly shortened recovery times and reduced complications:
- VATS (video-assisted thoracoscopic surgery): Smaller incisions, faster recovery
- Robotic-assisted surgery: Enhanced precision, reduced morbidity
- Equivalent cancer outcomes to open thoracotomy
Stage I patients who undergo successful surgery often require no further treatment — just ongoing surveillance.
SBRT: The Non-Surgical Alternative
Stereotactic body radiation therapy (SBRT) delivers high-dose, highly targeted radiation in 1-5 sessions. For medically inoperable early-stage patients, local control and survival outcomes rival surgery.
The landmark RTOG 0236 trial showed 97.6% 3-year primary tumor control and 90.6% local control for medically inoperable patients receiving 54 Gy in 3 fractions. A pooled analysis of two randomized trials comparing SBRT to lobectomy in operable patients found 95% 3-year overall survival for SBRT versus 79% for surgery.
SBRT is particularly valuable for:
- Patients with poor lung function
- Those with serious comorbidities precluding surgery
- Elderly patients preferring less invasive treatment
- Patients refusing surgery
Treatment completes in one week rather than requiring major surgery and 6-8 weeks recovery.
Proton Therapy: Precision Radiation
Proton beam therapy spares surrounding healthy tissue more effectively than conventional photon radiation. The physics advantage: protons deposit their energy at a specific depth (the Bragg peak) rather than passing through the body.
Dosimetric studies confirm that proton therapy significantly reduces radiation exposure to the lungs and heart compared to 3D-CRT, IMRT, and VMAT for stage I NSCLC. Survival data vs. modern photon techniques is still maturing, but the reduced collateral dose is a meaningful advantage in anatomically complex cases.
Proton therapy is most relevant for:
- Tumors near the heart, esophagus, or major airways
- Patients with already-compromised lung function who cannot tolerate additional scatter dose
- Cases where re-irradiation is needed and cumulative heart/lung dose is a concern
Nutritional and Metabolic Approaches to Lung Cancer Support
The Scientific Foundation: Cancer Cell Methionine Dependence
Cancer cells exhibit altered metabolism compared to healthy cells. They are disproportionately dependent on the amino acid methionine for growth and proliferation—a phenomenon called "methionine dependence" or the Hoffman effect.
When deprived of exogenous methionine, cancer cells arrest in late-S/G2 phase and undergo apoptosis, while normal cells survive through homocysteine recycling pathways. This creates a therapeutic window: restricting dietary methionine through a plant-based, low-methionine diet creates a metabolic environment less favorable for cancer cell survival.
Preclinical evidence supports this approach. In KRAS/Lkb1-mutant NSCLC mouse models, dietary methionine restriction decreased tumor progression and increased carboplatin treatment efficacy.
Plant-Derived Nutraceuticals
Specific natural compounds have been studied for their ability to target cancer cell vulnerabilities:
Curcumin - Has been investigated for its effects on cellular stress pathways in NSCLC cells through endoplasmic reticulum stress and mitochondrial membrane potential loss
EGCG (green tea catechin) - Inhibits PD-L1 expression in NSCLC cells via JAK2/STAT1 and EGFR/Akt signaling pathways
Sulforaphane - Attenuates EGFR signaling and promotes proteasomal degradation of EGFR, inhibiting tumor growth in EGFR-TKI-resistant NSCLC cells
These compounds work through different mechanisms than pharmaceutical drugs: affecting cellular health pathways being investigated in nutritional oncology research, reducing inflammatory signaling, and supporting immune function without systemic toxicity. This mechanistic diversity makes them useful complements to structured nutritional protocols.
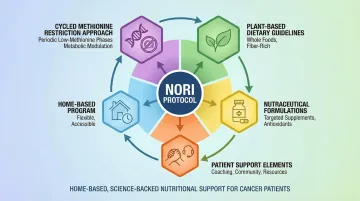
The NORI Protocol: A Structured Approach
The NORI Protocol is a science-based nutritional support program built on over 20 years of research. It combines cycled methionine restriction with proprietary nutraceutical formulations in a home-based format designed to work alongside or in place of conventional treatments.
The protocol targets universal cancer cell vulnerabilities rather than cancer-type-specific characteristics — and because it requires no clinic visits, it's accessible to patients worldwide.
Core components include:
- Cycled methionine restriction (typically 7 days on, 7 days off)
- Plant-based diet limiting protein to <0.3 g/kg body weight
- Fat intake restricted to <10% of calories
- Customized nutraceutical formulations manufactured in-house
- Continuous phone and Zoom support with NORI practitioners
Patients on the NORI Protocol report high quality of life—a critical consideration when weighing treatment options. For patients seeking to minimize toxic side effects while maintaining an active treatment approach, nutritional interventions offer a complementary path.
Observational Evidence
While lung-cancer-specific clinical trials for methionine restriction are lacking, observational data supports plant-based dietary patterns. Meta-analyses show that higher adherence to plant-based diets is associated with reduced all-cause and cancer-specific mortality in lung cancer patients.
Used alongside medical care, these nutritional strategies aim to make the internal environment less hospitable to cancer cells — without adding the toxic burden of systemic treatments.
Choosing and Combining Your Lung Cancer Treatment Plan
Biomarker Testing: The Essential First Step
Before committing to any treatment, comprehensive biomarker testing is non-negotiable. Results directly determine which chemotherapy-free options are available and which are most likely to work.
Required tests before first-line therapy:
- PD-L1 immunohistochemistry (IHC)
- Next-generation sequencing (NGS) panel covering EGFR, ALK, ROS1, BRAF, KRAS, MET, RET, and other actionable mutations
- Tumor mutational burden (TMB) when relevant
NCCN and ASCO guidelines strongly recommend broad molecular profiling prior to initiating first-line systemic therapy for advanced NSCLC. Yet real-world data shows that less than 50% of patients receive all key biomarker tests before starting treatment.
Pushing for complete testing before treatment begins helps ensure you and your care team are working with a full picture — not a partial one.
Integrative Treatment Planning
Many patients pursue conventional and nutritional strategies in parallel, using each to support the other. This kind of integrative planning can address both disease control and day-to-day quality of life.
Example combinations that work together:
- Immunotherapy + methionine restriction and nutraceuticals
- Targeted therapy + plant-based diet to support drug efficacy
- SBRT + nutritional support to enhance immune response
- Surgery + post-operative nutritional protocol to reduce recurrence risk
Discuss all planned interventions with your care team. Transparency prevents interactions and supports coordination. Most oncologists appreciate patients who take active roles in their care, though some may be unfamiliar with nutritional approaches.
For patients looking to explore nutritional approaches alongside conventional treatment, NORI offers a free initial consultation to discuss how the NORI Protocol might fit your situation. Reach out at 800-634-3804 or info@nutritionaloncology.net.
Frequently Asked Questions
Is lung cancer treatable without chemotherapy?
Yes, many lung cancer patients are treated without chemotherapy. Depending on stage and tumor biology, options include immunotherapy monotherapy, targeted therapy, surgery, or radiation as standalone approaches—particularly for early-stage or biomarker-selected patients. First-line pembrolizumab for patients with PD-L1 ≥50% is a prime example of effective chemotherapy-free treatment.
What is the 2 week rule for lung cancer?
The "2 week rule" (used primarily in the UK) refers to a clinical pathway where patients with suspicious symptoms—persistent cough, unexplained weight loss, or hemoptysis—are urgently referred for investigation within two weeks. This enables earlier diagnosis and treatment, improving outcomes.
What is targeted therapy for lung cancer and who qualifies?
Targeted therapy uses drugs matched to specific genetic mutations in the tumor (EGFR, ALK, KRAS G12C, ROS1, etc.). It's primarily available to NSCLC patients—typically adenocarcinoma—who test positive for a known actionable mutation through molecular profiling. Patients without identified mutations or with squamous histology are less likely to qualify.
Can nutrition and diet support lung cancer care?
Evidence supports dietary interventions—particularly low-methionine, plant-based diets combined with specific nutraceuticals—as a strategy to target cancer cell metabolic vulnerabilities and support the body's resilience during treatment. These are best used as complementary to, not replacements for, medical care.
How to deal with a lung cancer patient?
Prioritize active listening, help with practical tasks like appointments and medication management, and encourage engagement with the full range of treatment options—including supportive and nutritional care. Follow the patient's lead on how much information or involvement they want.
How to stop worrying about cancer?
Worry is a natural response—redirect it into action. Understanding your treatment options, optimizing nutrition, and connecting with others navigating similar experiences can shift anxiety into purposeful focus. Counseling support and mindfulness practices help too.
Treatment options for lung cancer have expanded considerably. Biomarker testing now opens the door to precision therapies with better tolerability profiles than traditional chemotherapy. Nutritional approaches can complement conventional care by targeting cancer cell metabolism directly. Start with comprehensive testing, make decisions with full information, and build a care team that respects the full range of your choices.
For personalized guidance on integrating nutritional support with your lung cancer care plan, contact NORI at 800-634-3804 or visit nutritionaloncology.net for free educational resources and weekly workshops.


