This article explains when breast cancer can be treated without chemotherapy, what the alternatives are, and how factors like tumor biology, stage, and genomic testing drive these decisions. The answer is never one-size-fits-all—it depends heavily on your cancer subtype, individual risk profile, and overall health. Let's walk through each of these dimensions so you can make an informed decision with your oncology team.
TLDR
- Up to 70% of early-stage HR+/HER2- breast cancer patients may avoid chemotherapy when genomic testing confirms low recurrence risk
- Tumor subtype, stage, lymph node status, and genomic scores determine if chemo is necessary
- Effective alternatives include surgery, hormone therapy, radiation, targeted drugs, and immunotherapy
- Methionine restriction and targeted nutraceuticals serve as evidence-informed integrative support alongside — or instead of — conventional treatment
- Patients who forgo chemotherapy in appropriate cases maintain similar survival rates with far better quality of life
Can You Really Treat Breast Cancer Without Chemotherapy?
Yes—skipping chemotherapy is not only possible but medically appropriate for a substantial portion of breast cancer patients, particularly those diagnosed with early-stage, hormone receptor-positive (HR+), HER2-negative tumors that haven't spread to lymph nodes. According to research from Fox Chase Cancer Center, approximately 70% of women with early-stage breast cancer can safely avoid chemotherapy.
Understanding Breast Cancer Subtypes
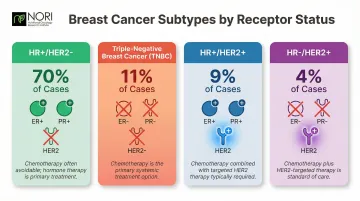
Breast cancer isn't a single disease—it's a collection of subtypes defined by hormone receptors and protein expression. Based on SEER data from 2018–2022, the four main subtypes are:
- HR+/HER2- (70% of cases): Most amenable to non-chemo treatment
- Triple-Negative (11%): Typically requires chemotherapy
- HR+/HER2+ (9%): Often needs chemotherapy plus targeted therapy
- HR-/HER2+ (4%): Usually requires chemotherapy and HER2-targeted drugs
The TAILORx Trial: A Landmark Shift
The TAILORx trial published in the New England Journal of Medicine studied 6,711 women with HR+/HER2-, node-negative breast cancer. Key findings:
- Women with mid-range Oncotype DX recurrence scores (11–25) showed no significant benefit from adding chemotherapy to hormone therapy
- At 9 years, distant recurrence rates were approximately 5% regardless of chemotherapy use
- Exception: Women under 50 with scores of 16–25 showed modest chemotherapy benefit
Important Distinctions
Not all breast cancers can skip chemotherapy. Triple-negative and certain HER2-positive cancers still typically require it. Additionally, there's a difference between:
- Completely avoiding chemotherapy (based on biology and genomic testing)
- Reducing chemotherapy intensity (shorter courses, lower doses, fewer drugs)
This decision should always be made with your oncologist, not alone.
What Factors Help Doctors Decide If Chemotherapy Is Necessary?
Your oncologist weighs multiple clinical and biological factors when recommending treatment. Here's what matters most:
Tumor Size and Stage
Small, localized tumors fully removable by surgery may not require systemic chemotherapy. Stage I and many Stage II cancers are often managed successfully without it, especially when other favorable factors are present.
NCCN guidelines note that for tumors up to 0.5 cm with no lymph node involvement, chemotherapy is generally not recommended.
Lymph Node Status
When cancer hasn't spread to lymph nodes, the case for skipping chemotherapy strengthens significantly. However, the RxPONDER trial showed that postmenopausal women with 1–3 positive nodes and low genomic scores can also safely forgo chemotherapy—a major expansion of who qualifies.
Hormone Receptors and Biomarkers
Your pathology report will identify:
- ER/PR status (estrogen/progesterone receptors): If positive, hormone therapy becomes a cornerstone alternative to chemo
- HER2 status: If positive, targeted therapy like trastuzumab may be used
- Ki-67: A proliferation marker indicating how fast cancer cells are dividing (though not yet standardized for treatment decisions per ASCO guidelines)
Genomic Testing: Predicting Chemotherapy Benefit
Oncotype DX and MammaPrint are FDA-cleared tests that analyze gene activity in tumor tissue to predict recurrence risk and chemotherapy benefit.
Oncotype DX produces a Recurrence Score (0–100):
- 0–25: Low to intermediate risk; most patients can skip chemotherapy
- 26–100: High risk; chemotherapy provides significant benefit
MammaPrint takes a different approach, assessing 70 genes to classify tumors as low or high genomic risk — and notably extends eligibility to some HR-negative patients that Oncotype DX doesn't cover.
Together, these tests have reshaped clinical practice. According to research published in Nature, Oncotype DX testing alone led to a 36% net reduction in chemotherapy recommendations in real-world practice.
Genomic scores don't operate in isolation, though. Biological factors — particularly menopausal status — determine how much weight those scores carry.
Menopausal Status
The RxPONDER trial revealed that menopausal status dramatically affects chemotherapy benefit. In women with 1–3 positive nodes and Recurrence Scores ≤25:
- Postmenopausal women: No benefit from chemotherapy
- Premenopausal women: Significant benefit from adding chemotherapy

Conventional Non-Chemo Treatment Options for Breast Cancer
When chemotherapy isn't necessary, several proven alternatives form the treatment foundation.
Surgery: Lumpectomy or Mastectomy
Surgical removal of the tumor is often the first and most critical step. Lumpectomy (breast-conserving surgery) removes the tumor and a margin of healthy tissue, preserving the breast. Mastectomy removes the entire breast and may be recommended when:
- Tumors are large relative to breast size
- Multiple tumor sites exist
- Margins cannot be cleared with lumpectomy
Surgery alone can be curative for very early-stage cancers when combined with appropriate follow-up care.
Radiation Therapy
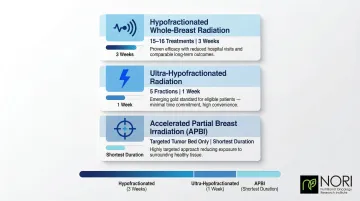
Radiation targets microscopic cancer cells remaining after surgery — especially after lumpectomy. Current approaches have shortened treatment timelines considerably:
- Hypofractionated whole-breast radiation: Shorter courses (15–16 treatments vs. 30+) with equivalent outcomes
- Ultra-hypofractionated regimens: The FAST-Forward trial demonstrated that 26 Gy in 5 fractions over 1 week is as effective as longer regimens
- Accelerated partial breast irradiation: For select low-risk patients, treating only the tumor bed rather than the whole breast
Hormone Therapy: The Cornerstone for HR+ Disease
For HR-positive breast cancer, hormone therapy blocks estrogen signals that fuel cancer growth—often replacing chemotherapy entirely.
The two main classes differ by menopausal status:
- Tamoxifen (premenopausal): Blocks estrogen receptors on cancer cells directly, taken daily for 5–10 years
- Aromatase inhibitors (postmenopausal): Letrozole, anastrozole, and exemestane stop estrogen production at the source — FDA-approved for both early-stage and metastatic HR+ disease
Targeted Therapy
These drugs attack specific cancer cell characteristics with far less toxicity than chemotherapy.
For HER2-positive breast cancer:
- Trastuzumab (Herceptin) and pertuzumab: Block HER2 protein signals that drive cancer growth
HR+/HER2-negative advanced disease has its own targeted class:
- CDK4/6 inhibitors (palbociclib, ribociclib, abemaciclib): Disrupt cancer cell division by blocking specific enzymes. Ribociclib received FDA approval in 2024 for adjuvant treatment of high-risk early breast cancer.
Immunotherapy: Breaking Ground in Triple-Negative Disease
While triple-negative breast cancer (TNBC) typically requires chemotherapy, immunotherapy is changing that equation. Pembrolizumab received FDA approval in 2021 for high-risk early TNBC combined with chemotherapy.
Even more promising: the BELLINI trial demonstrated that 33% of TNBC patients with high tumor-infiltrating lymphocytes achieved complete pathological response using immunotherapy alone—no chemotherapy. While immune-related side effects remain a concern, select TNBC patients may now have a viable path to treatment that avoids chemotherapy entirely.

Nutritional and Integrative Approaches: Expanding the Treatment Conversation
Beyond conventional medicine, emerging research reveals how diet and metabolic interventions can target cancer cell vulnerabilities. Cancer cells have distinct nutritional dependencies—particularly for the amino acid methionine—that can be therapeutically exploited.
Methionine Restriction: Targeting Cancer's Metabolic Weakness
Research shows that cancer cells are highly dependent on methionine (an essential amino acid) compared to normal cells. A methionine-restricted diet has been shown in preclinical studies to slow tumor growth and enhance other treatments by disrupting S-adenosylmethionine (SAM) biosynthesis, which cancer cells need for DNA methylation and proliferation.
In triple-negative breast cancer models, methionine restriction inhibited tumor growth, reduced metastasis, and created oxidative stress vulnerabilities that could be exploited therapeutically.
The NORI Protocol: A Science-Based Integrative Program
The Nutritional Oncology Research Institute (NORI) has developed a A 2018 case report documented a Stage IV ER+ breast cancer patient with bone metastasis who experienced bone pain subsiding within three months and a CA15-3 tumor marker drop from nearly 1,000 to 250 after starting the NORI Protocol alongside hormone therapy.
NORI offers free initial consultations at 800-634-3804 or info@nutritionaloncology.net to discuss how the program might fit your individual circumstances.
Clarifying the Role of Integrative Nutrition
That kind of documented response illustrates where integrative nutrition can make a meaningful difference — not as a standalone cure, but as an evidence-based addition to a broader treatment plan.
Nutritional protocols like NORI's work best alongside conventional care, supporting treatment tolerance, immune function, and quality of life. Always discuss integrative approaches with your oncologist to ensure safe, coordinated care.
How Genomic Testing Is Changing the Chemotherapy Decision
Genomic profiling has moved breast cancer treatment away from one-size-fits-all protocols toward precision medicine. Multi-gene expression assays like Oncotype DX and MammaPrint identify patients who gain little or no benefit from chemotherapy, reducing overtreatment on a large scale.
Impact on Chemotherapy Rates
SEER registry data from 2004–2015 shows that as Oncotype DX use increased from 1.5% to 34%, overall chemotherapy usage declined from 42% to 36%—preventing thousands of women from unnecessary treatment.
Understanding Test Results
Oncotype DX Recurrence Scores:
- 0–10: Excellent prognosis with hormone therapy alone
- 11–25: Minimal or no chemotherapy benefit for most patients (except premenopausal women with scores 16–25)
- 26–100: Significant chemotherapy benefit
MammaPrint:
- Low genomic risk: Chemotherapy unlikely to improve outcomes
- High genomic risk: Chemotherapy recommended
Eligibility Criteria
| Test | Target Population | Tumor Characteristics |
|---|---|---|
| Oncotype DX | HR+/HER2- | Stage I-IIIa; node-negative or 1-3 positive nodes |
| MammaPrint | HR+/HER2- or HR-/HER2- | Stage I-II; tumor ≤5.0 cm; 0-3 positive nodes |
The Bigger Picture
Genomic testing represents a broader shift toward treatment decisions rooted in tumor biology—targeting the specific vulnerabilities of your cancer rather than applying standard protocols. For patients with low recurrence scores, this means a legitimate, evidence-backed path to skipping chemotherapy entirely.
What to Know If You Choose to Skip or Reduce Chemotherapy
Patients have the right to make informed decisions about their own care, including declining chemotherapy. That decision deserves full information — including what the clinical data actually shows about survival when chemotherapy is skipped for the right candidates.
Survival Outcomes: The Evidence
For appropriate candidates, skipping chemotherapy doesn't compromise survival:
| Trial | Population | Hormone Therapy Alone | With Chemotherapy |
|---|---|---|---|
| TAILORx (9-year invasive disease-free survival) | Node-negative, Recurrence Score 11–25 | 83.3% | 84.3% |
| TAILORx (9-year overall survival) | Node-negative, Recurrence Score 11–25 | 93.9% | 93.8% |
| RxPONDER (5-year invasive disease-free survival) | Postmenopausal, 1–3 positive nodes, Recurrence Score 0–25 | 91.9% | 91.3% |
No statistically significant difference was found in either trial for these patient groups.
Quality of Life Benefits
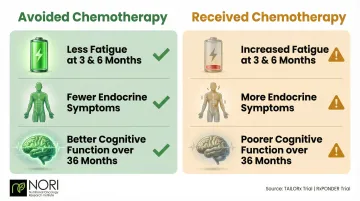
Women who avoided chemotherapy in these trials reported:
- Significantly less fatigue at 3 and 6 months (TAILORx patient-reported outcomes)
- Fewer endocrine symptoms during treatment
- Better cognitive function maintained over 36 months (RxPONDER cognitive substudy)
These findings matter practically: if your oncologist uses genomic testing such as Oncotype DX, the results can clarify whether you fall into a group where chemotherapy adds measurable benefit — or where hormone therapy alone delivers comparable outcomes with significantly less treatment burden.
Frequently Asked Questions
Is it possible to treat breast cancer without chemotherapy?
Yes, many patients—particularly those with early-stage, HR-positive/HER2-negative tumors and favorable genomic test results—can be effectively treated with surgery, hormone therapy, radiation, and/or targeted therapy without chemotherapy. Your specific cancer biology and genomic risk scores determine eligibility.
Will my life ever be the same after breast cancer?
Most patients treated for early-stage breast cancer, especially those who avoided chemotherapy's harsh side effects, report strong quality of life and return to normal activities. Ongoing monitoring stays important, but most women return to full, active lives.
What food to avoid after breast cancer?
Limit processed foods, high-sugar items, excessive alcohol, and red meat. A plant-rich, whole-food diet is broadly supported by oncology nutrition research for long-term survivorship.
At what stage of breast cancer is chemotherapy typically recommended?
Chemotherapy is most commonly recommended for Stage III–IV cancers, aggressive subtypes (triple-negative, certain HER2-positive), or when genomic testing indicates high recurrence risk. Many Stage I and Stage II patients successfully avoid it based on favorable biology and test scores.
What is the difference between hormone therapy and chemotherapy for breast cancer?
Chemotherapy kills rapidly dividing cells throughout the body, causing significant systemic side effects (hair loss, nausea, immune suppression). Hormone therapy specifically blocks estrogen signals that drive HR-positive cancer growth, making it a targeted, lower-toxicity option for eligible patients.
Breast cancer treatment has changed significantly. For many women, chemotherapy is no longer inevitable—genomic testing, hormone therapy, targeted drugs, and integrative nutritional approaches offer science-backed pathways that preserve quality of life without sacrificing survival outcomes. Understanding your cancer's unique biology is the starting point for exploring every option available to you.