
Introduction
Up to 80% of breast cancer patients now use complementary and alternative medicine (CAM). Chemotherapy-induced fatigue affects 72% of patients, and persistent side effects, treatment resistance, and gaps in how standard therapies address cancer stem cells have led many to seek evidence-based natural approaches alongside conventional care.
This article covers the most studied natural compounds and dietary strategies for breast cancer support — their biological mechanisms, and how patients can integrate them responsibly. The research draws on over 20 years of work in nutritional oncology, including protocols developed by NORI (Nutritional Oncology Research Institute) for patients navigating breast cancer at various stages.
TLDR
- Curcumin, EGCG, sulforaphane, resveratrol, and DIM demonstrate meaningful anti-breast-cancer activity in preclinical and clinical research
- Dietary strategies targeting estrogen metabolism, inflammation, and methionine restriction exploit known vulnerabilities in breast cancer cells
- For ER-positive breast cancer, several natural agents reduce aromatase activity and shift estrogen toward less potent forms
- Safe integration requires practitioner guidance — interactions with conventional treatments must be evaluated individually
Why Natural Therapies Are Gaining Ground in Breast Cancer Care
80% of breast cancer patients use some form of CAM, with many seeking relief from severe chemotherapy side effects including fatigue (72% experience it), immunosuppression, hair loss, and nausea. This isn't fringe behavior—it's mainstream reality driving scientific investigation.
Conventional-only approaches have documented limitations:
- Multidrug resistance develops in many breast cancers over time
- Chemotherapy side effects damage quality of life
- Cancer stem cells remain largely untargeted by standard therapies, enabling relapse and metastasis
- Treatment failure leaves patients with few options
Understanding how natural therapies fit into a treatment plan requires one key distinction.
Complementary therapy means using natural approaches alongside evidence-based conventional treatment to manage side effects, improve quality of life, and potentially enhance outcomes. Alternative therapy means using natural approaches instead of conventional treatment.
The former has a legitimate and growing evidence base. The latter carries real risk when substituted for proven interventions—particularly in early-stage disease where conventional treatment offers high cure rates.
Evidence-Based Natural Compounds for Breast Cancer Support
Curcumin: Enhancing Chemotherapy While Managing Risks
Curcumin, the active compound in turmeric, demonstrates multiple health-supporting mechanisms:
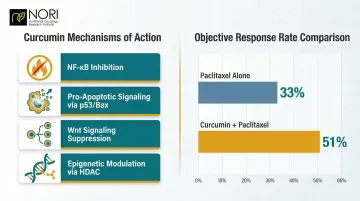
- NF-κB inhibition blocks inflammatory signaling that promotes tumor growth
- Pro-apoptotic signaling via p53/Bax pathways affects cellular pathways, as investigated in nutritional oncology research
- Wnt signaling suppression targets breast cancer stem cells
- Epigenetic modulation through HDAC inhibition alters gene expression
A randomized, placebo-controlled trial showed that intravenous curcumin combined with paclitaxel improved objective response rates: 51% versus 33% in advanced breast cancer patients. Apoptosis in triple-negative breast cancer (TNBC) cells also increased when both agents were combined, compared to paclitaxel alone.
One practical limitation: oral curcumin absorbs poorly. Specialized formulations — liposomal, micronized, or combined with piperine — measurably improve bioavailability.
Before using curcumin, disclose it to your oncology team. It exhibits antiplatelet activity, acting as a blood thinner, which creates serious risks before surgery or alongside anticoagulants like warfarin.
EGCG: Epigenetic Reactivation and Tamoxifen Synergy
For patients on tamoxifen, EGCG (epigallocatechin gallate from green tea) stands out for a specific reason: it appears to work with endocrine therapy rather than against it. Its mechanisms include:
- Reactivates silenced tumor suppressor genes via epigenetic mechanisms, particularly the SCUBE2 gene hypermethylated in breast cancer
- Inhibits COX-2 expression, reducing inflammatory signaling
- Has been investigated for its effects on cellular stress pathways in both ER-positive and ER-negative cell lines
- Increases tamoxifen bioavailability without interfering with its active metabolite (endoxifen)
A randomized pharmacokinetic study confirmed that green tea extract (300 mg EGCG/day) does not negatively alter endoxifen levels in breast cancer patients taking tamoxifen. This makes EGCG especially notable as a safe adjuvant support compound for patients on endocrine therapy.
EGCG combined with other demethylating compounds like sulforaphane shows epigenetic protective effects, correcting aberrations in estrogen receptor-negative cell lines and may sensitize ER-negative tumors to tamoxifen in cell line models.
Sulforaphane: Targeting Cancer Stem Cells
Sulforaphane, concentrated in broccoli sprouts, targets one of the harder-to-reach aspects of tumor biology — cancer stem cells:
- Targets breast cancer stem cells by downregulating Wnt/β-catenin self-renewal pathways
- Has been investigated for its effects on cellular stress pathways through PARP-1 and caspase activation
- Causes epigenetic repression of hTERT (the enzyme enabling cancer cell immortality)
A Johns Hopkins presurgical trial assigned 30 postmenopausal breast cancer patients to either broccoli sprout extract (200 μmol isothiocyanates/day) or placebo for two weeks before surgery. Results trended toward increased apoptosis (cleaved caspase-3) and decreased proliferation (Ki-67), with urinary proteomic analysis confirming apoptosis induction and immune modulation.
Sulforaphane at 40 μM also suppressed β-catenin expression in MDA-MB-231 breast cancer cells in a time-dependent manner, disrupting the Wnt pathway critical for cancer stem cell survival.
Resveratrol: Aromatase Inhibition with Bioavailability Hurdles
Resveratrol from grapes and red wine demonstrates dual activity:
- Aromatase inhibitor: Suppresses CYP19 promoter transactivation, reducing estrogen production
- Estrogen metabolism modulator: Shifts metabolism toward less potent forms
- COX-2 suppression: Reduces inflammatory signaling
- PI3K pathway inhibition: Blocks cancer cell survival signals
- Epigenetic effects: DNMT inhibition demethylates tumor suppressor genes
A 12-week supplementation study (50 mg twice daily) reduced DNA methylation of the RASSF1A tumor suppressor gene in breast tissue of women at increased risk.
The central challenge with resveratrol is delivery. It has extremely low water solubility and metabolizes rapidly, which limits how much actually reaches target tissue. Studies comparing standard powder to soluble formulations show substantially higher plasma levels with specialized versions. Micronized or liposomal delivery systems are generally required for clinical relevance.
DIM: Favorable Estrogen Shifts But Endoxifen Concerns
Unlike EGCG, DIM (3,3'-diindolylmethane from cruciferous vegetables) carries a significant caution for tamoxifen users — though its estrogen-modulating effects are otherwise promising:
- Modulates aromatase expression, reducing estrogen production
- Inhibits angiogenesis, starving tumors of blood supply
- Sensitizes HER-2/neu-expressing cancer cells to herceptin
- Enhances tamoxifen chemosensitivity in randomized trial data
A randomized, placebo-controlled trial (98 women completed) showed that 300 mg/day BR-DIM significantly increased the 2:16α-hydroxyestrone ratio (+3.2 for DIM versus -0.7 for placebo, P < 0.001). Serum SHBG also rose with DIM (+25 ± 22 nmol/L) compared to placebo (+1.1 ± 19 nmol/L). These are favorable estrogen-related shifts.

The same trial, however, found that plasma tamoxifen metabolites — including endoxifen, 4-OH tamoxifen, and N-desmethyl-tamoxifen — were significantly reduced in the BR-DIM group versus placebo (P < 0.001). Since endoxifen is tamoxifen's active metabolite, this reduction may meaningfully compromise endocrine therapy efficacy.
If you are on tamoxifen, exercise caution with concentrated DIM supplements. Whole cruciferous vegetables deliver DIM precursors at food-level doses without the concentrated amounts that appear to interfere with tamoxifen metabolism.
Dietary Strategies That Target Breast Cancer Biology
Anti-Inflammatory Dietary Patterns
Chronic inflammation drives breast cancer progression through the arachidonic acid/COX-2 pathway. An anti-inflammatory diet rich in omega-3 fatty acids, colorful vegetables, and fiber—while low in refined carbohydrates and processed foods—can reduce this risk.
A pooled analysis of 31 studies found that adherence to the Mediterranean diet was associated with a 13% reduction in breast cancer risk (HR: 0.87, 95% CI: 0.82–0.92). Postmenopausal women experienced a 12% significant reduction (HR: 0.88; 95% CI: 0.84, 0.92).
Cruciferous Vegetables as a Dietary Cornerstone
Broccoli, cabbage, cauliflower, and Brussels sprouts provide precursors (I3C, glucosinolates) that convert to active compounds (DIM, sulforaphane) in the gut during digestion.
A meta-analysis found that high cruciferous vegetable intake was significantly associated with reduced breast cancer risk (RR = 0.85, 95% CI = 0.77–0.94).
Regular intake at food levels—not just supplement doses—provides meaningful cancer-preventive and tumor-suppressing effects by modulating estrogen metabolism and targeting cancer stem cells.
Soy and Phytoestrogens: Navigating the Nuance
Many breast cancer patients worry about soy and ER+ disease, but the evidence tells a different story.
A large pooled analysis of 9,514 survivors found that post-diagnosis soy intake (≥10 mg isoflavones/day) reduced breast cancer recurrence by 26% (HR = 0.74, 95% CI = 0.60–0.92), particularly among postmenopausal and ER-positive women. For ER+ disease specifically, soy protein and products were inversely associated with cancer-specific mortality (HR = 0.75, 95% CI = 0.60–0.92).
Genistein, the primary soy isoflavone, acts as a selective estrogen receptor modulator with context-dependent effects. The practical takeaways:
- Whole soy foods (tofu, edamame, tempeh) at 1–2 servings daily appear safe and potentially protective
- The concern applies specifically to high-dose isolated isoflavone supplements in postmenopausal women—avoid these without medical guidance
- Both the American Cancer Society and AICR actively encourage whole soy food consumption for breast cancer survivors
Methionine Restriction: A Targeted Nutritional Strategy
Beyond phytoestrogens, another dietary lever targets cancer at the metabolic level. Cancer cells have an elevated dependence on the amino acid methionine—a vulnerability known as "methionine dependence" or the Hoffman Effect. Normal cells can synthesize methionine from other compounds when dietary intake is low; cancer cells cannot adapt and become selectively stressed.
Restricting dietary methionine—primarily found in animal proteins, eggs, and certain plant foods like nuts and seeds—creates selective metabolic stress in cancer cells. Cancer cells are known to have elevated reactive oxygen species (ROS) levels — pro-oxidants are being investigated for how they may interact with these cellular conditions. Methionine restriction's effects on glutathione and cellular stress pathways are being studied in nutritional oncology research, with ongoing investigation into how different cell types respond.
NORI's protocol, developed over 20 years of focused research, combines cycled methionine restriction—alternating periods of strict restriction with modified intake—with nutraceutical combinations being studied for their role in oxidative stress pathways and cellular health. Natural agents used in this approach include high-dose Vitamin C, sodium selenite, and vitamin K3—compounds also being explored alongside conventional care approaches in ongoing nutritional oncology research.
Early-phase clinical trials examining methionine restriction alongside established therapies have reported favorable safety profiles and tolerability, supporting its use as an adjunct nutritional strategy.
Avoiding Dietary Factors That Promote Breast Cancer Growth
Three well-documented dietary risk factors have direct mechanistic links to breast cancer progression:
- Excess body fat increases breast cancer-specific mortality by 22% (RR: 1.22, 95% CI: 1.13–1.32). In postmenopausal women, adipose tissue drives circulating estrogen levels through aromatase enzyme activity in fat cells.
- Alcohol raises risk linearly at 9% per 10g/day (roughly one standard drink). Even less than one drink daily increases risk by 4% (RR: 1.04, 95% CI: 1.01–1.07), through elevated estrogen and acetaldehyde-related DNA damage.
- High-glycemic diets increase breast cancer risk by 8% (RR: 1.08, 95% CI: 1.02–1.14). Women in the highest glycemic load quintile had a 31% higher risk of breast cancer-specific mortality and 26% higher all-cause mortality—driven by insulin/IGF-1 signaling that promotes tumor growth.

Naturally Reducing Estrogen: What the Research Shows
Why Estrogen Reduction Matters for Breast Cancer
Approximately 70-80% of breast cancers are hormone receptor-positive (ER+), meaning excess estrogen—particularly estradiol produced via the aromatase enzyme—fuels tumor growth. Natural aromatase inhibitors represent a well-studied area, with multiple plant compounds showing activity comparable in mechanism (though not always in potency) to pharmaceutical aromatase inhibitors.
Natural Aromatase Inhibitors from Food and Plants
Several food-derived compounds have demonstrated measurable aromatase-inhibiting activity in research:
- Resveratrol suppresses CYP19 promoter transactivation, reducing aromatase gene expression and blocking androgen-to-estrogen conversion
- Biochanin A (from red clover) directly blocks aromatase activity and reduces aromatase mRNA expression in MCF-7 breast cancer cells
- EGCG (from green tea) inhibits aromatase enzyme activity through multiple pathways
- White button mushrooms (Agaricus bisporus) contain unsaturated fatty acids—including linoleic, linolenic, and conjugated linoleic acid—that interact directly with aromatase active site regions to inhibit conversion
DIM and Estrogen Metabolism
DIM promotes the conversion of estrone to 2-hydroxyestrone (a less proliferative estrogen metabolite) rather than 16-alpha-hydroxyestrone (a more potent, pro-cancer metabolite). This shift in the 2:16 ratio is measurable and stands among the better-documented natural mechanisms for estrogen management in ER+ breast cancer.
A randomized trial by Dalessandri et al. (2004) documented a significant favorable shift in this ratio with 300 mg/day BR-DIM supplementation in women at elevated breast cancer risk.
Lifestyle Factors That Reduce Estrogen Levels Naturally
Four modifiable lifestyle factors have demonstrated measurable effects on circulating estrogen:
- Body weight: Adipose tissue is a primary site of peripheral estrogen production via aromatase. Weight loss in overweight or obese postmenopausal women directly lowers circulating estrogen levels.
- Physical exercise: Systematic reviews show exercise reduces circulating estrone and estradiol while increasing SHBG, which binds estrogen and reduces its bioavailability. A meta-analysis of 8 studies (n = 1,482) found a borderline significant estradiol reduction (SMD = −0.09, 95% CI [−0.20, 0.01], p = 0.07).
- Alcohol restriction: Even moderate alcohol consumption raises estrogen levels. Eliminating or strictly limiting intake produces measurable hormonal benefits.
- Stress and sleep: Chronic stress and poor sleep alter hormonal balance through cortisol-to-estrogen signaling pathways. Consistent sleep and stress management are practical, low-cost components of estrogen control.

Safety and Integration: What Patients Need to Know
"Natural" Is Not Synonymous with "Risk-Free"
Natural compounds can cause serious harm when used incorrectly:
- Curcumin acts as a blood thinner, creating dangerous bleeding risks before surgery or when combined with anticoagulants
- High-dose soy supplements may be contraindicated in ER+ cases, despite whole soy foods being safe
- St. John's Wort strongly induces CYP3A4, reducing chemotherapy drug levels by up to 42% (demonstrated with irinotecan, where plasma concentrations of the active metabolite SN-38 decreased by 42%)
- High-dose antioxidants may reduce radiation therapy effectiveness if taken at the wrong time, as radiotherapy works partly through ROS-mediated cancer cell death
Critical Rule: Patients must disclose all supplements to their oncology team. Non-disclosure creates serious safety risks.
The Case for Integrative or Nutritional Oncology Guidance
The growing complexity of natural-conventional interactions, combined with the genuine promise of nutritional medicine, makes professional guidance a practical necessity — not a precaution to skip.
The Society for Integrative Oncology (SIO) Clinical Practice Guidelines offer the only comprehensive evidence-based framework for incorporating complementary therapies into conventional oncology, and ASCO endorses them. Working with a trained integrative or nutritional oncology practitioner gives patients access to:
- Personalized supplementation timing that avoids interference with active radiation or chemotherapy
- Appropriate dosing guidance — distinguishing food-level intake from concentrated supplement doses
- Compound combinations that work together without triggering dangerous interactions
NORI's Home-Based Nutritional Support Program
NORI's Home-Based Nutritional Support Program
For patients looking for that kind of structured, personalized guidance, NORI is one science-grounded option. The program provides:
- Free initial consultations to discuss your specific situation
- Unlimited support through phone and Zoom consultations
- Home-based format requiring no clinic visits, making it accessible worldwide
- Personalized care tailored to individual cancer type, stage, and treatment plan
NORI's work draws on founder Mark Simon's research beginning in 2004, centered on cycled methionine restriction and nutraceutical combinations that target metabolic patterns common across cancer cell types. All nutraceuticals are formulated and manufactured in-house, with verified potency, purity, and quality.
Contact NORI at 800-634-3804 or info@nutritionaloncology.net to schedule your free consultation.
Frequently Asked Questions
How can I lower my estrogen naturally for breast cancer?
Key approaches include:
- Consuming dietary aromatase inhibitors: resveratrol, DIM from cruciferous vegetables, white button mushrooms
- Maintaining a healthy weight to reduce peripheral estrogen production via adipose tissue
- Limiting alcohol and exercising regularly
These should complement — not replace — prescribed hormonal therapy like tamoxifen or aromatase inhibitors.
Can natural therapies replace conventional breast cancer care?
Natural therapies cannot replace proven conventional cancer care for most patients. They do have well-documented roles as complementary tools — improving outcomes, reducing side effects, and supporting quality of life under medical supervision. Early-stage breast cancer carries high cure rates with conventional cancer care, and foregoing that carries significant risk.
Is curcumin safe to take during chemotherapy?
While curcumin has shown chemosensitizing effects with paclitaxel (improving response rates in clinical trials), it acts as a blood thinner and may interact with certain chemotherapy drugs. Oncologist consultation is essential before use, as timing and dosage are critical variables that must be individualized.
Are soy products safe for women with ER-positive breast cancer?
Whole soy foods (tofu, edamame, tempeh) consumed in moderate amounts (1-2 servings daily) appear safe and may be protective based on population studies showing 26% reduced recurrence risk. High-dose isolated isoflavone supplements require more caution and individual medical evaluation, particularly for postmenopausal women on aromatase inhibitors.
What is methionine restriction and how does it relate to breast cancer?
Cancer cells require disproportionately high methionine because they depend on glutathione to suppress their already-elevated ROS levels. Restricting dietary methionine depletes this defense, affecting cellular pathways, as investigated in nutritional oncology research — the "Hoffman Effect" — while healthy cells adapt through alternative protective pathways. It is an active area of nutritional oncology research with a clear mechanistic rationale.
What is the difference between complementary and alternative therapy in breast cancer?
Complementary therapies are used alongside conventional treatment to manage side effects, improve quality of life, and potentially enhance treatment outcomes. Alternative therapies are used instead of conventional treatment. Most evidence-based natural approaches fall into the complementary category, as replacing proven conventional treatments — especially in early-stage disease — carries significant risk.


